NIAID plays a unique role in the global effort against malaria in that it funds the majority of basic malaria research. The NIAID Malaria Research Program encompasses a broad range of topics, covering the full cycle of malarial disease—from parasite to mosquito to human host. Increased knowledge of these three elements and the multifaceted interactions among them is critical in developing effective tools to prevent and control malaria.
Parasite Biology
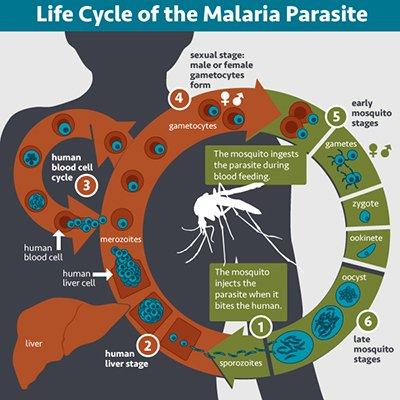
Life cycle of the malaria parasite. View a full size image.
Malaria can be caused by several species of Plasmodium parasites, each of which has a complex life cycle (see illustration). Research in recent decades has shed light on many aspects of Plasmodium biology, broadening understanding of how parasites interact with the human immune system, cause human disease, and are transmitted by mosquitoes. Still, in these fundamental areas and others, important questions remain unanswered and new questions have arisen. NIAID supports basic research on parasite biology to unravel the complexities of such crucial processes and increase knowledge of disease transmission, parasite immune evasion, and the emergence of drug resistance.
Vector Biology
Malaria parasites are transmitted to human hosts by female mosquitoes of the genus Anopheles. A diverse group of Anopheles (30 to 40 species) serves as vectors of human disease. Several physiological, behavioral, and ecological characteristics determine how effective various Anophelesspecies are as vectors of malaria. NIAID supports research on such characteristics to better understand the parasite-vector relationship and determine why some mosquito species transmit malaria parasites while others do not.
Disease Pathogenesis
Malaria pathogenesis is the process by which malaria parasites cause illness, abnormal function, or damage in their human hosts. “Uncomplicated” malaria entails a series of recurring episodes of chills, intense fever, and sweating and sometimes includes other symptoms such as headache, malaise, fatigue, body aches, nausea, and vomiting.
In some cases, and especially in groups such as children and pregnant women, the disease can progress to “severe malaria,” including complications such as cerebral malaria/coma, seizures, severe anemia, respiratory distress, kidney and liver failure, cardiovascular collapse, and shock. Long-term impacts include death, disability, and significant socioeconomic burden on societies where the disease is prevalent. A better understanding of the biological processes underlying the progression of infection to disease is urgently needed to reduce the morbidity and mortality of malaria.
Genomics
Over the past serveral years, the completion of several genome projects related to malaria has marked the beginning of a new era of malaria research. NIAID-supported researchers have sequenced the genomes of 16 Anopholine mosquito species, including the Anopheles gambiae mosquito, a major malaria vector, and more than 100 isolates of both Plasmodium falciparum, the deadliest malaria parasite, and Plasmodium vivax, the most widespread malaria parasite. These accomplishments, in conjunction with National Institutes of Health-funded sequencing of the human genome, have provided scientists with unprecedented information on complete sets of human, parasite, and mosquito genes. Through such advances, as well as ongoing and planned genomics projects on additional parasites and vectors, NIAID hopes to supply scientists with the tools needed to identify targets for effective disease interventions.
Immunology
The interaction between the Plasmodium parasite and host immune system during infection strikes a tenuous balance. The relationship can elicit protective immunity or trigger harmful immune responses. The complex nature of both the malaria parasite and the human immune response has made it difficult to unravel the mechanisms of protection or pathology in humans. An improved understanding of the immunology of malaria is likely to provide key insights into ways to enhance human immunity while reducing disease pathogenesis.
Epidemiology
Epidemiological data is critical to both developing novel vaccines and drugs and implementing effective control and prevention programs. Understanding malaria on a population level and determining the biological, behavioral, and environmental factors that influence malaria epidemiology and transmission are especially important as the global community strengthens anti-malaria efforts.
Scientific Advances
NIAID Researchers Study Causes of Brain Swelling in Cerebral Malaria
September 13, 2023In children with cerebral malaria, brain swelling can cause seizures, coma, and death. Researchers from NIAID and their colleagues studied children with cerebral malaria in Malawi to better understand the underlying causes of these devastating symptoms.
NIAID-Funded Study Traces Evolution of Malaria Drug Resistance in E. Africa
August 24, 2023A new NIAID-funded study shows how inconsistent malaria control measures in East Africa could be aiding the emergence of drug-resistant mutations in the primary parasite that causes malaria.