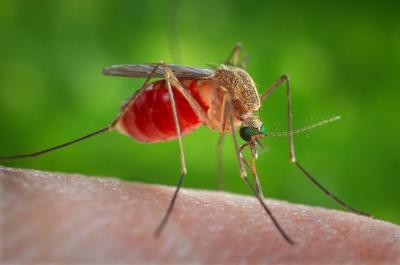
This is an enlarged view of a Culex quinquefasciatus mosquito that had landed upon the skin of a human host, and was about to insert its needle-sharp proboscis through the skin, which would enable it to obtain its blood meal.
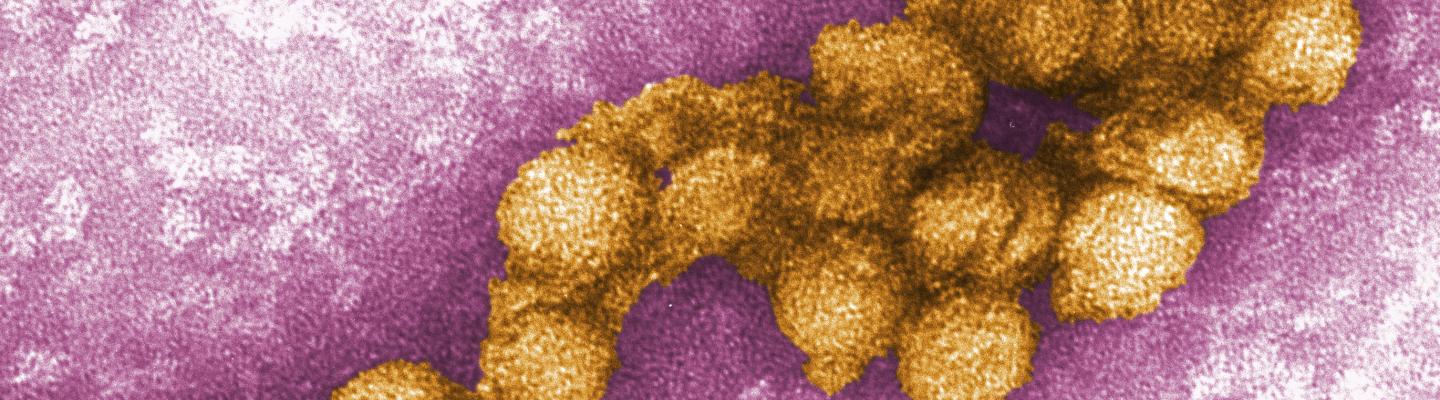
West Nile virus (WNV) is a member of the flavivirus family, which includes other mosquito-borne viruses such as dengue and Zika. WNV emerged for the first time in the Western Hemisphere in New York City in 1999 and has since spread across the United States. It is now the most common arthropod-borne virus found in the U.S.
Most people (about 8 out of 10) who are infected with WNV develop no symptoms at all, while a smaller proportion develop mild symptoms of fever, body aches, skin rash, and swollen lymph nodes. If the virus crosses the blood-brain barrier, however, it can cause life-threatening conditions that include inflammation of the brain and spinal cord. There are no specific therapies for serious WNV infection and there is currently no vaccine available to prevent infection.
NIAID supports research aimed at better understanding of the interactions between WNV virus, mosquitoes, and the human immune response, as well as broad-spectrum anti-flavivirus drug discovery efforts.
NIAID Now Blog
Funded Research News
Related Public Health and Government Information
To learn about risk factors for WNV and current prevention and treatment strategies visit the MedlinePlus West Nile virus site.