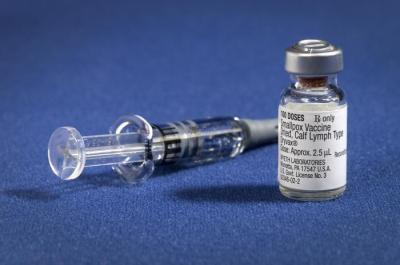
Vaccinia (smallpox) vaccine, derived from calf lymph, and currently licensed in the United States, is a lyophilized, live-virus preparation of infectious vaccinia virus. It does not contain smallpox (variola) virus.
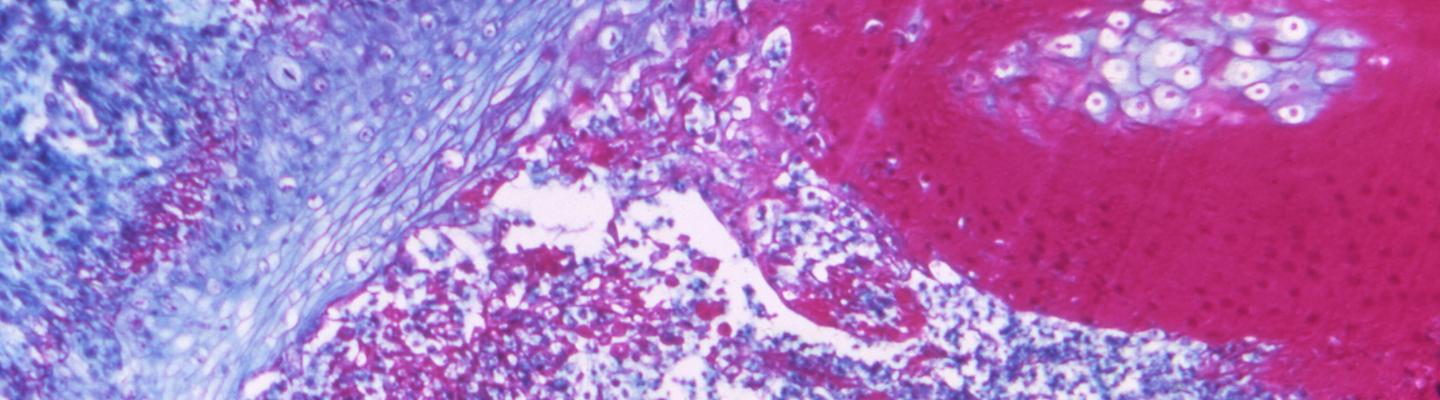
Smallpox, caused by the variola virus, was a highly contagious infectious disease that caused infected individuals to develop a fever and a progressive, disfiguring skin rash. Three of out 10 individuals infected with smallpox died. Many survivors have permanent scars, often on their faces, or were left blind. Through vaccination, the disease was eradicated in 1980. However, research for effective vaccines, drugs and diagnostics for smallpox continues in the event it is used as a bioterror weapon.
The last naturally occurring case of smallpox was reported in 1977. In 1980, the World Health Organization declared that smallpox had been eradicated. Currently, there is no evidence of naturally occurring smallpox transmission anywhere in the world. Although a worldwide immunization program eradicated smallpox disease decades ago, small quantities of smallpox virus officially still exist in two research laboratories in Atlanta, Georgia, and in Russia.
Why Is the Study of Smallpox a Priority for NIAID?
Smallpox is a category A pathogen which are those organisms/biological agents that pose the highest risk to national security and public health because they can be easily disseminated or transmitted from person to person, result in high mortality rates and have the potential for major public health impact, might cause public panic and social disruption, and require special action for public health preparedness.
How Is NIAID Addressing This Critical Topic?
NIAID supports basic, preclinical, and clinical research needed to advance product development for biodefense and emerging infectious diseases. Product development goals in this arena have shifted from a “one bug-one drug” approach to a more flexible strategy that is applicable to a broad spectrum of infectious diseases. Specifically, this broad-spectrum approach is being used to develop products effective against a variety of pathogens and toxins; find technologies that can be widely applied to improve multiple classes of products; and establish platforms that can reduce the time and cost of creating new products. This is evident in both the treatment and vaccine research NIAID has supported for smallpox, outlined below.
Funded Research News
Related Public Health and Government Information
To learn about risk factors for smallpox and current prevention and treatment strategies visit the MedlinePlus smallpox site.