NIAID supports basic research to understand the biology of microbes, their behavior and how they develop resistance and cause disease.
How Microbes Develop Resistance and Cause Disease
Some microbes have structural characteristics that make them inherently resistant to certain antimicrobials. Microbes that once were susceptible to a particular drug also can develop resistance through genetic mutations, by acquiring resistance genes from other organisms and through complex biochemical mechanisms. Learn more about the causes of antimicrobial resistance.
NIAID research aims to better understand the fundamental biology of microbes and how they can block antimicrobial drugs. This knowledge can help scientists identify novel drug targets, find new ways to exploit old targets, and develop novel diagnostics and vaccines. For example, recent NIAID-supported research demonstrated the genetic basis for how N. gonorrhoeae can develop resistance to the common antibiotic azithromycin. Researchers found that N. gonorrhoeae can acquire resistance from other Neisseria species, and demonstrated that the bacteria serve as a reservoir for resistance genes. The findings provide a better understanding of the genetic basis of resistance and may have implications on the development of improved diagnostics and surveillance efforts.
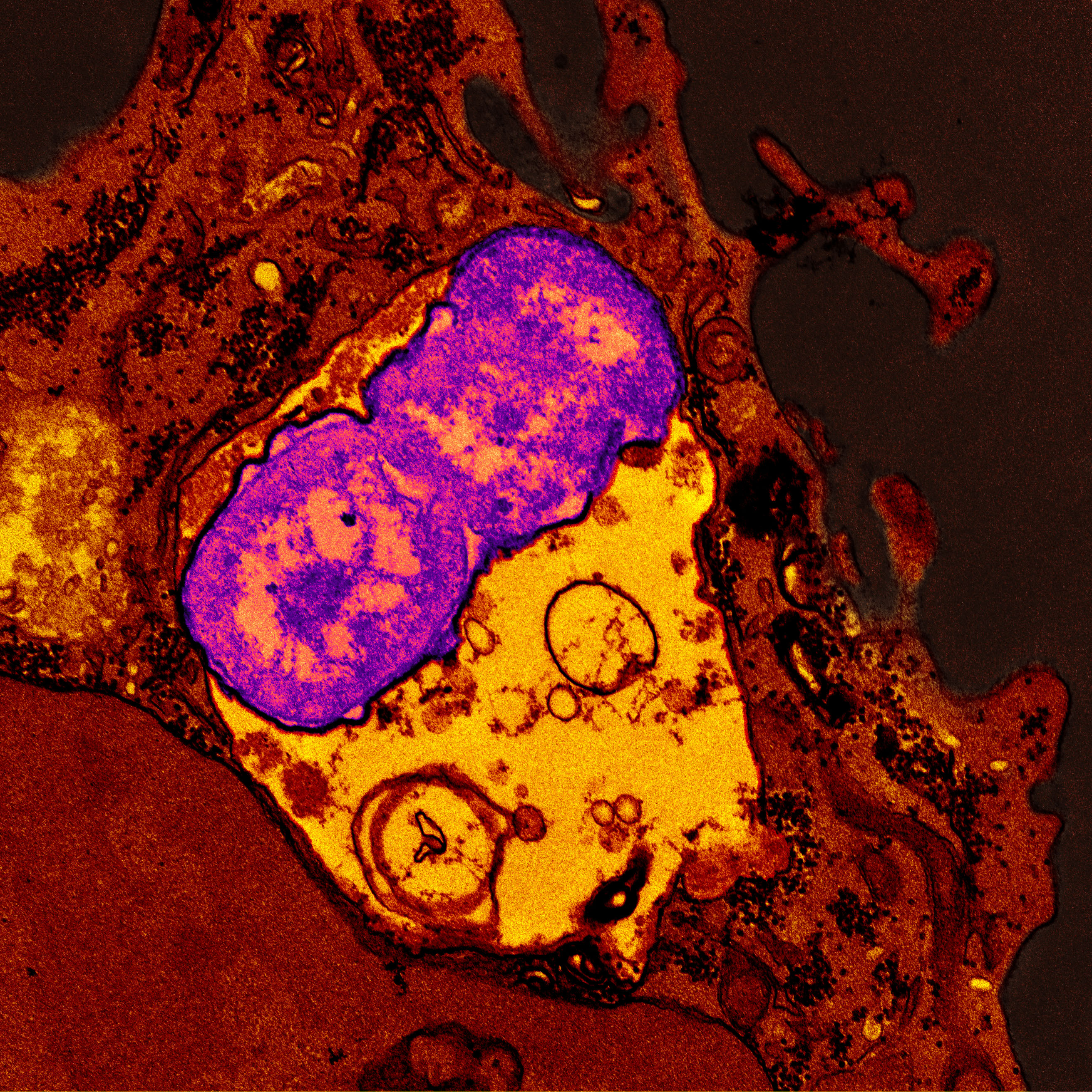
Microbial pathogenesis, or how microbes cause disease, is another important area of investigation. Researchers supported by NIAID are conducting studies on how microbes colonize the host, which toxins microbes produce, and how microbes avoid or overcome an attack by the host’s immune defenses. Understanding and identifying contributors to bacterial virulence, including toxins, can lead to potential therapeutic targets. Drugs designed to target virulence factors could avoid selective pressure and should limit disruption to the microbiome.
Host-Pathogen Interactions
Understanding host-pathogen interactions can help scientists design effective treatments. NIAID researchers also are exploring host factors—the traits that may influence an individual’s susceptibility and response to disease. Host-targeted interventions that harness a person’s immune system, rather than the microbes themselves, are being explored as alternative treatments to conventional antimicrobial drugs. NIAID scientists are working on an antibody against carbapenem-resistant Klebsiella pneumoniae that is designed to boost the killing of bacteria by a type of white blood cell.
Genomics and Computational Biology Approaches
Genome-based analyses can reveal important insights for combatting antimicrobial resistance. By isolating and comparing the genomes of the same species of microbe from different geographic locations or from different human populations, scientists can identify when and where resistance first emerged and specific mechanisms of resistance. Much of this research was not even feasible just a decade ago. NIAID’s Genomic Centers for Infectious Diseases uses comparative genomics to evaluate strains, species and clinical samples.
The NIAID-funded Bacterial and Viral Bioinformatics Resource Center includes bacterial species and offers scientists genome assembly, annotation and analysis, as well as RNA sequencing, and interactive phylogenetic trees (showing the evolutionary relationships among organisms), among other services. NIAID initiated the BRCs to collect, archive, update and integrate a variety of research data and make available critical analytical resources, bioinformatics tools, workspaces and services for data analysis.
Scientific Advances
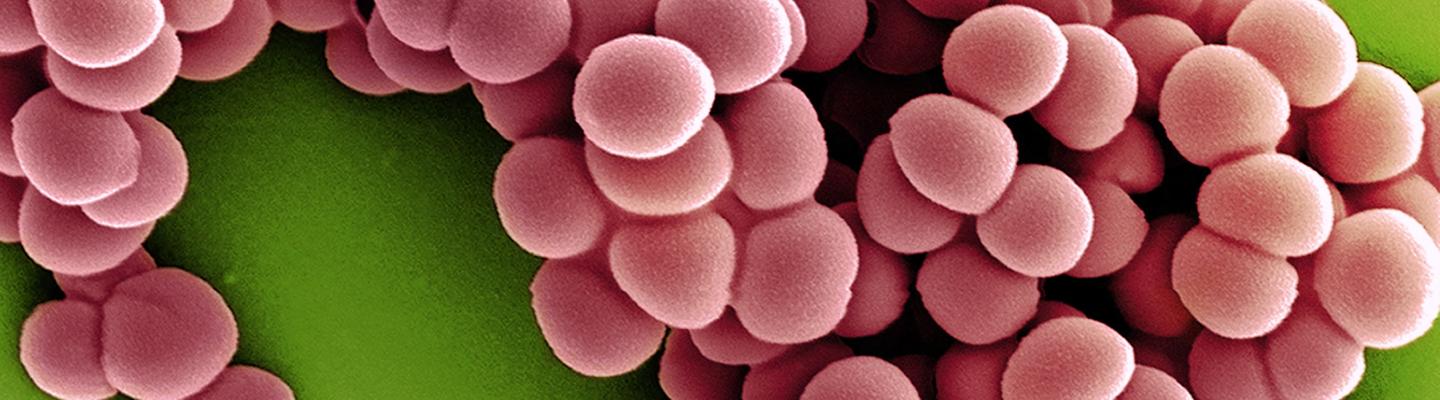
NIH Investigates Multidrug-Resistant Bacterium Emerging in Community Settings
September 6, 2023New “hypervirulent” strains of the bacterium Klebsiella pneumoniae have emerged in healthy people in community settings, prompting a National Institutes of Health research group to investigate how the human immune system defends against infection.