Antibodies are made by B cells, and each antibody recognizes a unique molecule. This specificity makes antibodies a powerful research, diagnostic, and therapeutic tool. Antibodies are used by researchers for a variety of reasons, but they are mainly used for labeling, in one form or another. For instance, scientists studying a protein, like a cytokine, need a way to isolate this protein from samples. With an antibody specific for the protein, they may easily separate the protein through binding assays.
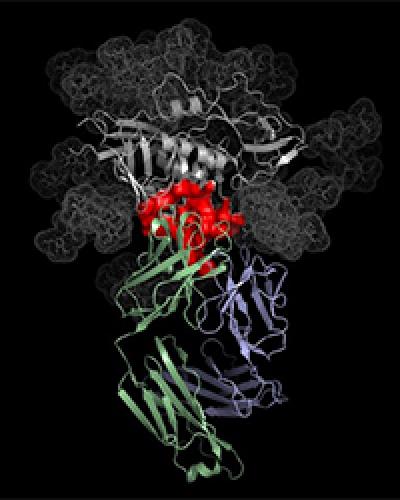
VRC01 antibody (blue and green) binding to HIV (grey and red).
Scientists also may want to visualize a protein on a cell to understand how it works. Antibodies coupled with other tags, like a fluorescent label, allow researchers to image a cell and see how the protein is expressed. This concept also is useful for separating cells. For example, scientists studying T cells need to isolate T cells from all others. By identifying proteins expressed only on T cells, an antibody specific for this protein allows researchers to pull T cells out of a sample.
Antibodies are used clinically and are the subject of numerous clinical trials. Conceptually, antibodies may be used to soak up or block harmful proteins in a disease setting. For instance, tumor necrosis factor (TNF) is an inflammatory cytokine that may worsen symptoms in several diseases, like Crohn's disease and rheumatoid arthritis. There are several Food and Drug Administration-approved drugs that target TNF, and these drugs are actually antibodies that block TNF and alleviate disease symptoms. Antibodies also are used in cancer therapy. Proteins that suppress immune cells may be targeted by antibodies, so that immune cells remain active to clear away cancer cells.
Genetic Engineering
Genetic engineering is another useful research tool. In model systems, scientists can study and manipulate genes, in animals ranging from fruit flies to mice, to understand what causes various diseases and how to treat them. Gene therapy has been studied in clinical trials to treat diseases. For instance, patients with genetic immune disorders, like severe combined immunodeficiency (SCID), may receive a copy of the gene they are missing, to potentially restore healthy immune function. Gene therapy studies are ongoing, to understand long-term effectiveness and side effects.
Epigenetics
Epigenetics is the study of gene expression, particularly factors unrelated to changes in gene sequence that may turn it "on" or "off." Genes are DNA blueprints that code for RNA, which is translated into protein, like a cytokine. When an immune cell activates and needs to produce cytokines to signal other cells, the cytokine gene needs to be in an "on" state for this to occur. If the gene is in an "off" state, it cannot be stimulated, no matter what cues are present around the cell.
Scientists study how gene status is regulated through epigenetic processes that alter the on/off state, generally by regulating how tightly DNA is packed around proteins in the nucleus. Tightly wound DNA is inaccessible and "off," while loosely wound DNA is accessible and "on." Understanding epigenetic mechanisms may lead to new therapy, and drugs that target epigenetic processes are currently being studied in clinical trials. Instead of blocking a harmful protein with antibodies, a drug that targets epigenetic mechanisms may potentially prevent that harmful protein from being made in the first place.
Immunotherapy
Immunotherapy is the manipulation of the immune system to solve a health problem. There are many clinical trials examining immunotherapy for cancer by directing the patient's own immune system to attack cancer cells. Researchers have examined the potential of innate and adaptive cells to fight cancer. Similar to how vaccines induce memory responses against a particular microbe, cancer immunotherapy is an attempt to induce effective immune responses against a specific cancer.
Immunotherapy may be used to treat food allergy, another focus of clinical trials. Oral immunotherapy is feeding small, increasing amounts of a food allergen—milk, egg, or peanut—to an allergic person over time. Studies show that this can decrease sensitivity to an allergen, but it must be done under the supervision of a physician. New clinical trials are testing other routes, like a skin patch, to deliver immunotherapy for allergies.
Metabolism
In recent years, scientists have focused on the role of the immune system in regulating metabolism. The activity of immune cells, like macrophages, may influence a variety of diseases including diabetes, obesity, and atherosclerosis. For example, signals secreted by macrophages may affect the activity and size of adipocytes, which are the cells that store fat as energy. More research must be done to understand how the immune system and inflammatory responses contribute to metabolic disorders.
Microbiome
The human microbiome is the trillions of relatively harmless microorganisms (bacteria, fungi, and viruses) that reside on and in the human body. These resident microbes also are referred to as commensals. Scientists are beginning to understand the essential role of the microbiome in human health. Without commensals, the immune system fails to develop properly.
Studies in animal models have shown that commensals play an important role in altering the activity of immune cells, which may lead to different outcomes in disease settings. More work is needed however, to understand how different commensals skew immune responses and how these changes may affect various human diseases.
Resources
Scientific Advances
Scientists Developed a System of Organoids That Can Model Activity in B Cells
November 6, 2024A system of organoids (lab-grown, miniature organ mimics) that can model activity in immune cells called B cells. The organoids produced antibodies in response to influenza vaccines and act as a model for B cell-related disorders.
Study Reveals How Young Children’s Immune Systems Tame SARS-CoV-2
October 13, 2023A study of infants and young children found those who acquired SARS-CoV-2 had a strong, sustained antibody response to the virus and high levels of inflammatory proteins in the nose but not in the blood. This immune response contrasts with that typically seen in adults with SARS-CoV-2 infection.
Understanding the Immune Response to the Fungus Cryptococcus in Healthy People
May 20, 2015NIAID researchers describe the immune responses of healthy people who developed the fungal infection cryptococcosis.