Microbes, such as bacteria, viruses, fungi, and parasites, are living organisms that evolve over time. Their primary function is to reproduce, thrive, and spread quickly and efficiently. Therefore, microbes adapt to their environments and change in ways that ensure their survival. If something stops their ability to grow, such as an antimicrobial, genetic changes can occur that enable the microbe to survive. There are several ways this happens.
Natural (Biological) Causes
Selective Pressure
In the presence of an antimicrobial, microbes are either killed or, if they carry resistance genes, survive. These survivors will replicate, and their progeny will quickly become the dominant type throughout the microbial population.
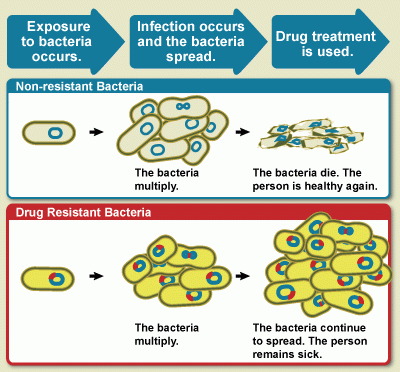
Diagram showing the difference between non-resistant bacteria and drug resistant bacteria. Non-resistant bacteria multiply, and upon drug treatment, the bacteria die. Drug resistant bacteria multiply as well, but upon drug treatment, the bacteria continue to spread.
Mutation
Most microbes reproduce by dividing every few hours, allowing them to evolve rapidly and adapt quickly to new environmental conditions. During replication, mutations arise and some of these mutations may help an individual microbe survive exposure to an antimicrobial.
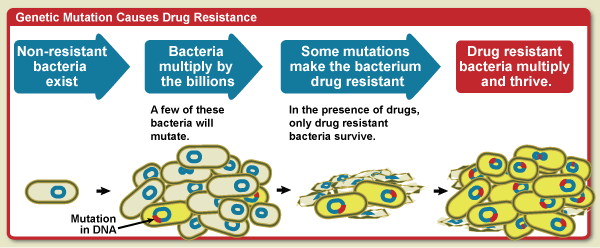
Diagram showing that when bacteria mulitply some will mutate. Some of those mutations can make the bacteria resistance to drug treatment. In the presence of the drugs, only the resistant bacteria survive and then multiply and thrive.
Gene Transfer
Microbes also may get genes from each other, including genes that make the microbe drug resistant. Bacteria multiply by the billions. Bacteria that have drug-resistant DNA may transfer a copy of these genes to other bacteria. Non-resistant bacteria receive the new DNA and become resistant to drugs. In the presence of drugs, only drug-resistant bacteria survive. The drug-resistant bacteria multiply and thrive.
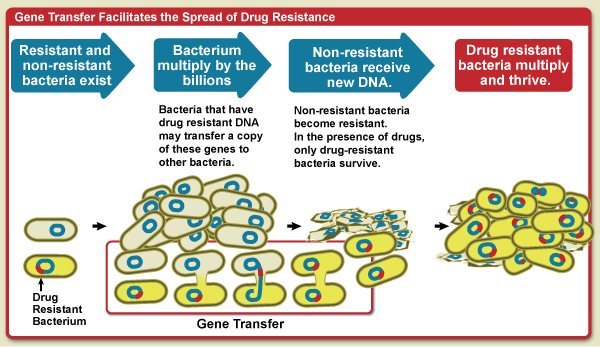
Diagram showing how gene transfer facilitates the spread of drug resistance. Bacteria multiply by the billions. Bacteria that have drug resistant DNA may transfer a copy of these genes to other bacteria. Non-resistant bacteria recieve the new DNA and become resistant to drugs. In the presence of drugs, only drug-resistant bacteria survive. The drug resistant bacteria multiply and thrive.
Societal Pressures
The use of antimicrobials, even when used appropriately, creates a selective pressure for resistant organisms. However, there are additional societal pressures that act to accelerate the increase of antimicrobial resistance.
Inappropriate Use
Selection of resistant microorganisms is exacerbated by inappropriate use of antimicrobials. Sometimes healthcare providers will prescribe antimicrobials inappropriately, wishing to placate an insistent patient who has a viral infection or an as-yet undiagnosed condition.
Inadequate Diagnostics
More often, healthcare providers must use incomplete or imperfect information to diagnose an infection and thus prescribe an antimicrobial just-in-case or prescribe a broad-spectrum antimicrobial when a specific antibiotic might be better. These situations contribute to selective pressure and accelerate antimicrobial resistance.
Hospital Use
Critically ill patients are more susceptible to infections and, thus, often require the aid of antimicrobials. However, the heavier use of antimicrobials in these patients can worsen the problem by selecting for antimicrobial-resistant microorganisms. The extensive use of antimicrobials and close contact among sick patients creates a fertile environment for the spread of antimicrobial-resistant germs.
Agricultural Use
Scientists also believe that the practice of adding antibiotics to agricultural feed promotes drug resistance. More than half of the antibiotics produced in the United States are used for agricultural purposes.1, 2 However, there is still much debate about whether drug-resistant microbes in animals pose a significant public health burden.
References
- National Research Council, Committee on Drug Use in Food Animals. The use of drugs in food animals: benefits and risks. Washington (DC): National Academy Press; 1999.
- Mellon M, Benbrook C, Benbrook KL. Hogging it: Estimates of antimicrobial abuse in livestock. Cambridge (MA): Union of Concerned Scientists; 2001.