36 Results
Measuring Innovation: Laboratory Infrastructure to Deliver Essential HIV Clinical Trial Results
HIV clinical trials network laboratory functions will continue to evolve to align with scientific priorities and research approaches.
Some People with Advanced HIV Have Anti-CD4 Autoantibodies Associated with Dampened Immune Recovery
A NIAID-sponsored analysis found that people with advanced HIV and anti-CD4 autoantibodies experienced limited CD4+ T-cell reconstitution through up to four years of observation after ART initiation, highlighting a potential immune effect of long-term unsuppressed HIV. The findings were published in Clinical Infectious Diseases.

Shaping the Next Era of HIV Therapeutics and Care
The Institute aims to foster the next generation of discoveries that will enable people with HIV to experience a typical lifespan with high life quality. Scientific priorities include removing the chronic HIV medication burden; reducing the incidence of concurrent TB and hepatitis; and ensuring scientific advances can feasibly be scaled to all who stand to benefit.
HIVR4P 2024 Research Highlights: Reproductive Health While on PrEP and Signals to Guide HIV Vaccines and Cure
New NIAID-supported science presented at the 2024 HIV Research for Prevention conference in Lima, Peru features a breadth of HIV discovery and translational findings and enriches the evidence base on HIV pre-exposure prophylaxis within the context of reproductive health.
Defining the Goals of HIV Science Through 2034
Discovery, Development and Delivery for an Increasingly Interconnected HIV Landscape
By Carl Dieffenbach, Ph.D., director, Division of AIDS, NIAID
We envision an HIV research enterprise that follows a logical evolution in addressing new scientific priorities informed by previous research progress. We will fund our next networks to align with updated research goals to take us through the end of 2034. Our capacity must reflect an increasing interdependence across clinical practice areas and public health contexts.
Study Links Certain Vaginal Bacteria and Inflammatory Marker to Increased Odds of Acquiring HIV Among Cisgender Women
Fourteen vaginal bacterial species and the presence of a protein that promotes inflammation were associated with increased odds of HIV acquisition in a study of more than 500 cisgender women in African countries with high HIV incidence. The study was the largest to date to prospectively analyze the relationship between both the vaginal microbiome and vaginal tissue inflammation and the likelihood of acquiring HIV among cisgender women in this population.
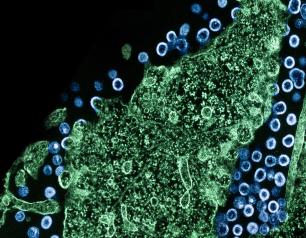
Bringing HIV Study Protocols to Life with Representative, High-Quality Research
The HIV clinical trials network sites have made tremendous contributions to NIH’s scientific priorities by offering direct access to and consultation with populations most affected by HIV globally, and by delivering high-quality clinical research with strong connections to trusted community outreach platforms. Future networks will need to maintain core strengths of current models while expanding capacity in areas vital to further scientific progress. These include operations that inform pandemic responses and extending our reach within communities impacted by HIV, including populations historically underrepresented in clinical research.
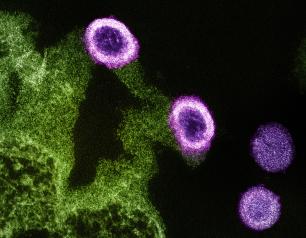
Charting the Path to an HIV-Free Generation
NIAID supports four research networks as part of its HIV clinical research enterprise. Every seven years, the Institute engages research partners, community representatives, and other public health stakeholders in a multidisciplinary evaluation of network progress toward short- and long-term scientific goals. Pregnancy, childbirth and the postnatal period are a key focus of NIAID HIV research and call for measures to support the health of people who could become pregnant as well as their infants.
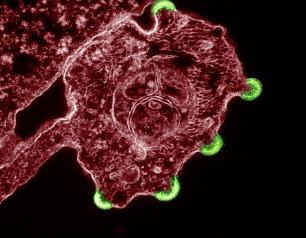
Broadly Neutralizing Antibodies Evaluated in Many HIV Cure Strategies
Many promising HIV cure strategies use broadly neutralizing antibodies, or bNAbs, which can neutralize a wide range of HIV variants, homing in on and binding to specific viral components, and then acting to destroy the virus by triggering an immune response. Several HIV bNAbs have been developed and tested to determine whether they can prevent or treat HIV. NIAID and partners are evaluating bNAb-based strategies alone and in combination with other immunity-enhancing strategies for HIV clearance in clinical trials in in Africa, North and South America, and Southeast Asia.

NIAID Research to Eliminate the Threat of Viral Hepatitis Across the Globe
Viral hepatitis affects the lives of about one in twenty people in the world, resulting in over a million deaths each year. NIAID is working on many ways to prevent and treat the different types of hepatitis, including the development of vaccines and improved therapeutics and diagnostics. July 28 is observed annually as World Hepatitis Day, providing an opportunity to reflect on the impact of hepatitis on global health and focus on strategies to reduce its burden. To observe World Hepatitis Day, NIAID highlights recent advancements researchers have made in these areas.
AIDS 2024: NIH Research Updates, Inequities, U=U, and Doxy PrEP (VIDEO)
The AIDS 2024 conference is taking place in Munich, Germany. HIV.gov is providing daily coverage.

Exploring the Potential of Pharmacies to Increase Access to HIV Testing
In 2022, an estimated 31,800 people were diagnosed with HIV in the United States. Although HIV incidence has declined by 12% from 2018 to 2022, many people are still undiagnosed due to limited access to HIV testing resources.

Our Words Have Power—NIAID Embraces Respectful, Inclusive, and Person-First Language
by Jeanne Marrazzo, M.D., M.P.H., NIAID Director
Our institute is responsible not only for advancing scientific knowledge, but for doing so in a way that honors the dignity, individuality, and autonomy of the people affected by the health issues we address. For this reason, I am very proud to share the updated NIAID HIV Language Guide, a thoroughly vetted resource to inform our written and verbal communications.
Antiretroviral Drug Improves Kidney Function After Transplant in People with HIV
An HIV drug that suppresses the activity of the CCR5 receptor—a collection of proteins on the surface of certain immune cells—was associated with better renal function in kidney transplant recipients with HIV compared to people who took a placebo in a randomized trial. According to the authors, these findings warrant further exploration of the benefit of CCR5 antagonists in all kidney transplant recipients regardless of HIV status. The findings of the NIAID-sponsored trial were presented today at the 2024 American Transplant Congress in Philadelphia.
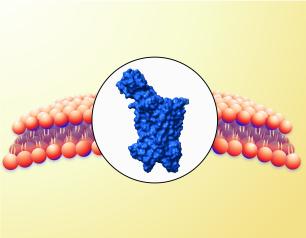
Proof-of-Concept Study Shows an HIV Vaccine Can Generate Key Antibody Response in People
An HIV vaccine candidate elicited trace levels of HIV broadly neutralizing antibodies and high levels of other key immune cells in an early-stage clinical trial. This immune response is an important signal that, if antibody levels can be further amplified, the vaccination strategy might be able to prevent HIV.
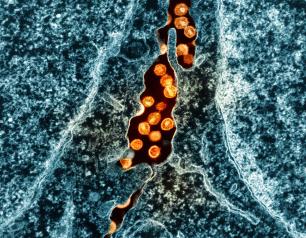
NIAID Marks HIV Vaccine Awareness Day 2024
On the 27th observance of HIV Vaccine Awareness Day (Saturday, May 18), we express our gratitude to the dedicated global community of scientists, advocates, study participants, study staff, and funders working toward a safe, effective, durable, and accessible HIV vaccine.

The HIV Field Needs Early-Stage Investigators (VIDEO)
by Jeanne Marrazzo, M.D., M.P.H., NIAID Director
The HIV research community is led by scientists with deep personal commitments to improving the lives of people with and affected by HIV. Our collective decades of work have generated HIV testing, prevention and treatment options beyond what we could have imagined in the 1980s. Those advances enable NIAID to explore new frontiers: expanding HIV prevention and treatment modalities, increasing understanding of the interplay between HIV and other infectious and non-communicable diseases, optimizing choice and convenience, and building on the ever-growing knowledge base that we need to develop a preventive vaccine and cure. The next generation of leaders will bring these concepts to fruition, and we need to welcome and support them into the complex and competitive field of HIV science.
Sexually Transmitted Infections—A Closer Look at NIAID Research
Sexually transmitted infections (STIs) are caused by bacteria, viruses, or parasites. NIAID supports research across the spectrum from basic to clinical science to develop effective diagnostic, preventive and therapeutic approaches to STIs in alignment with the National STI Strategic Plan. In recognition of National STI Awareness Week, NIAID shares a snapshot of new projects and recent scientific advances in STI research.

NIH Ending the HIV Epidemic Projects Bridge Gaps Between HIV Research and Public Health Practice (VIDEO)
The National Institutes of Health recently issued $26M in awards to HIV research institutions in its fifth year supporting implementation science under the Ending the HIV Epidemic in the U.S. initiative. These awards are the latest investments in a program that is rapidly and rigorously generating evidence to inform the unified domestic HIV response by agencies in the Department of Health and Human Services.
HIV Treatment Research and Key Takeaways: Dr. Dieffenbach’s Final Update from CROI 2024 (VIDEO)
On March 6 as the 2024 Conference on Retroviruses and Opportunistic Infections (CROI) was winding down, HIV.gov spoke with Carl Dieffenbach, Ph.D., director of NIAID's Division of AIDS, about highlights of long-acting HIV treatment research discussed at the conference. He spoke with Brian Minalga, M.S.W., deputy director of the NIH-supported Office of HIV/AIDS Network Coordination. Watch their conversation.

Centering Women and Girls’ Health in HIV Research
Women account for approximately 23 percent of people with HIV in the United States. In recent years, women aged 25 to 34 comprised the highest number of new diagnoses. Furthermore, Black women, transgender women, and women aged 13 through 24 are more likely to experience health disparities associated with lack of access to HIV testing, treatment, and prevention resources. This weekend marked National Women and Girls HIV/AIDS Awareness Day. NIAID supports research programs that focus on HIV and other health outcomes in women to inform and enable more targeted and effective HIV prevention, care, and treatment.

Promising Outcomes with HIV Treatment Started Promptly After Birth: Deborah Persaud Presents at CROI 2024 (VIDEO)
On the final day of the 2024 Conference on Retroviruses and Opportunistic Infections (CROI), HIV.gov spoke with Deborah Persaud, M.D., professor of Pediatrics at the Johns Hopkins University School of Medicine and director of the Division of Pediatric Infectious Diseases at Johns Hopkins Children's Center, who reported findings from a study about whether very early initiation of antiretroviral therapy (ART) may limit the establishment of HIV reservoirs in newborns, potentially enabling ART-free remission. Dr. Persaud spoke with Catey Laube of NIAID's Office of Communications and Government Relations. Watch their conversation.

Addressing Liver and Cardiovascular Disease among People with HIV and HIV Prevention During Pregnancy: Dr. Dieffenbach’s Second Update from CROI 2024 (VIDEO)
On Tuesday at the 2024 Conference on Retroviruses and Opportunistic Infections (CROI), HIV.gov spoke with Carl Dieffenbach, Ph.D., director of NIAID’s Division of AIDS, about research on common health complications of HIV and the safety of an HIV prevention tool during pregnancy. He spoke with Miss Molly Moon, M.S.W., Deputy Director of the NIH-supported Office of HIV/AIDS Network Coordination. Watch their conversation.

Doxy-PEP, HIV Vaccines and Community-Engaged Research: Discussions with Carl Dieffenbach and LaRon Nelson at CROI 2024 (VIDEO)
During the first full day of presentations at the 2024 Conference on Retroviruses and Opportunistic Infections, HIV.gov spoke with Carl Dieffenbach, Ph.D., director of NIAID’s Division of AIDS, and LaRon Nelson, Ph.D., R.N., F.N.P., F.N.A.P., F.N.Y.A.M., F.A.A., professor and associate dean at the Yale School of Nursing. They discussed Doxy-PEP for STI prevention, HIV vaccines, community engagement in research, and more. Watch their discussions.

NIAID’s Dr. Jeanne Marrazzo Previews CROI 2024 (VIDEO)
HIV.gov opened their coverage of the 2024 Conference on Retroviruses and Opportunistic Infections (CROI) today with a conversation with NIAID Director Jeanne Marrazzo, M.D., M.P.H. She spoke with Miguel Gomez, director of HIV.gov about what she’s looking forward to hearing about at the conference, how the studies presented at research conferences ultimately get translated into clinical practice or HIV prevention or treatment services, and the important roles of early-career investigators. Watch their conversation with Dr. Marrazzo.


