43 Results
NIH Statement on World AIDS Day
December 2, 2024
Together with our partners, the National Institutes of Health (NIH) commemorates World AIDS Day and affirms our commitment to bolstering the extraordinary gains achieved in HIV science and to persevering until we end HIV-related illness and stigma. As we mark this observance, we celebrate the people who enable scientific progress, honor the loved ones and leaders we have lost, and reflect on the work that remains to ensure the health and life quality of all people affected by, and living with, HIV.
NIH Trial of Rectal Microbicide for HIV Prevention Begins in the United States
October 31, 2024
A clinical trial has launched to examine the safety and acceptability of a novel rectal HIV microbicide douche containing the antiretroviral drug tenofovir. While HIV incidence is slowly decreasing in the United States, 67% of U.S. HIV diagnoses from 2018-2022 were among gay, bisexual, and other men who have sex with men, pointing to the need for expanded HIV prevention options. The mid-stage study is sponsored by the National Institutes of Health’s National Institute of Allergy and Infectious Diseases and implemented through the NIH-funded HIV Prevention Trials Network.
Kidney Transplantation Between Donors and Recipients with HIV Is Safe
October 16, 2024
Kidney transplantation from deceased donors with HIV to recipients with HIV was safe and comparable to kidney transplantation from donors without HIV.
An Isolated Viral Load Test May Generate False Positive Results for People Using Long-Acting PrEP
July 23, 2024
A single laboratory-based HIV viral load test used by U.S. clinicians who provide people with long-acting, injectable cabotegravir (CAB-LA) HIV pre-exposure prophylaxis (PrEP) did not reliably detect HIV in a multi-country study. In the study, a single positive viral load test was frequently found to be a false positive result.
Long-Acting Injectable Cabotegravir for HIV Prevention Is Safe in Pregnancy
July 23, 2024
Long-acting injectable cabotegravir (CAB-LA) was safe and well tolerated as HIV pre-exposure prophylaxis (PrEP) before and during pregnancy in the follow-up phase of a global study among cisgender women.
Exploratory Analysis Associates HIV Drug Abacavir with Elevated Cardiovascular Disease Risk in Large Global Trial
July 23, 2024
Current or previous use of the antiretroviral drug (ARV) abacavir was associated with an elevated risk of major adverse cardiovascular events (MACE) in people with HIV, according to an exploratory analysis from a large international clinical trial primarily funded by the National Institutes of Health (NIH).
NIH Statement on Preliminary Efficacy Results of Twice-Yearly Lenacapavir for HIV Prevention in Cisgender Women
June 26, 2024
The injectable antiretroviral drug lenacapavir was safe and 100% effective as long-acting HIV pre-exposure prophylaxis (PrEP) among cisgender women in a Phase 3 clinical trial, according to top-line findings released by Gilead Sciences, Inc., the study sponsor. Lenacapavir is administered every six months, making it the most durable HIV prevention method to have shown efficacy in this population.
U.S. Clinical Trials Begin for Twice-Yearly HIV Prevention Injection
June 4, 2024
Two clinical trials have launched to examine a novel long-acting form of HIV pre-exposure prophylaxis (PrEP) in cisgender women and people who inject drugs.
Novel Vaccine Concept Generates Immune Responses that Could Produce Multiple Types of HIV Broadly Neutralizing Antibodies
May 30, 2024
Using a combination of cutting-edge immunologic technologies, researchers have successfully stimulated animals’ immune systems to induce rare precursor B cells of a class of HIV broadly neutralizing antibodies (bNAbs). The findings, published today in Nature Immunology, are an encouraging, incremental step in developing a preventive HIV vaccine.
Children Surpass a Year of HIV Remission after Treatment Pause
March 6, 2024
Four children have remained free of detectable HIV for more than one year after their antiretroviral therapy (ART) was paused to see if they could achieve HIV remission, according to a presentation today at the 2024 Conference on Retroviruses and Opportunistic Infections (CROI) in Denver.

Long-Acting HIV Treatment Benefits Adults with Barriers to Daily Pill Taking and Adolescents with Suppressed HIV
March 6, 2024
Long-acting, injectable antiretroviral therapy (ART) suppressed HIV replication better than oral ART in people who had previously experienced challenges taking daily oral regimens and was found safe in adolescents with HIV viral suppression, according to two studies presented today at the 2024 Conference on Retroviruses and Opportunistic Infections (CROI) in Denver. Both studies were sponsored by the National Institute of Allergy and Infectious Diseases (NIAID), part of the National Institutes of Health, in collaboration with other NIH institutes.
Semaglutide Reduces Severity of Common Liver Disease in People with HIV
March 5, 2024
A weekly injection of semaglutide was safe and reduced the amount of fat in the liver by 31% in people with HIV and metabolic dysfunction-associated steatotic liver disease (MASLD), according to a presentation today at the 2024 Conference on Retroviruses and Opportunistic Infections (CROI) in Denver. This is the first clinical trial of semaglutide for MASLD in people with HIV.

Vaginal Ring and Oral Pre-Exposure Prophylaxis Found Safe for HIV Prevention Throughout Pregnancy
March 5, 2024
The monthly dapivirine vaginal ring and daily oral pre-exposure prophylaxis (PrEP) with tenofovir disoproxil fumarate and emtricitabine were each found to be safe for HIV prevention among cisgender women who started using one of them in their second trimester of pregnancy, according to findings presented today at the 2024 Conference on Retroviruses and Opportunistic Infections (CROI) in Denver.
Tools Underestimate Cardiovascular Event Risk in People with HIV
March 4, 2024
The elevated cardiovascular disease risk among people with HIV is even greater than predicted by a standard risk calculator in several groups, including Black people and cisgender women, according to analyses from a large international clinical trial primarily funded by the National institutes of Health and presented at the 2024 Conference on Retroviruses and Opportunistic Infections (CROI) in Denver.
Statement: Long-Acting HIV Treatment Demonstrates Efficacy in People with Challenges Taking Daily Medicine as Prescribed
February 21, 2024
Long-acting antiretroviral therapy (ART) with cabotegravir and rilpivirine was superior in suppressing HIV replication compared to daily oral ART in people who had been unable to maintain viral suppression through an oral daily regimen, according to interim data from a randomized trial. Upon review of these findings, an independent Data and Safety Monitoring Board (DSMB) recommended halting randomization and inviting all eligible study participants to take long-acting ART.
NIH-Developed HIV Antibodies Protect Animals in Proof-of-Concept Study
January 17, 2024
Three different HIV antibodies each independently protected monkeys from acquiring simian-HIV (SHIV) in a placebo-controlled proof-of-concept study intended to inform development of a preventive HIV vaccine for people. The antibodies—a human broadly neutralizing antibody and two antibodies isolated from previously vaccinated monkeys—target the fusion peptide, a site on an HIV surface protein that helps the virus fuse with and enter cells.
NIH Research Identifies Opportunities to Improve Future HIV Vaccine Candidates
December 14, 2023
An effective HIV vaccine may need to prompt strong responses from immune cells called CD8+ T cells to protect people from acquiring HIV, according to a new study from researchers at the National Institute of Allergy and Infectious Diseases (NIAID), part of the National Institutes of Health, and colleagues. The study findings, appearing in Science, draw comparisons between the immune system activity of past HIV vaccine study participants and people with HIV who naturally keep the virus from replicating even in the absence of antiretroviral therapy (ART).
NIH Clinical Trial of Tuberculous Meningitis Drug Regimen Begins
December 7, 2023
A trial of a new drug regimen to treat tuberculous meningitis (TBM) has started enrolling adults and adolescents in several countries where tuberculosis (TB) is prevalent. The trial will include 330 participants aged 15 years and older who have or are likely to have TBM based on signs and symptoms, including people living with and without HIV. Because pregnant women are eligible to enroll in this study with appropriate consent, a small number of pregnant women are expected to be included.
World AIDS Day 2023
December 1, 2023
On this 35th World AIDS Day, the National Institutes of Health (NIH) joins its partners in honoring the lives lost due to the HIV pandemic. For decades, this virus has exacted a tragic toll, affecting people, families, and communities worldwide, threatening social and economic development, and exacerbating stigma, often toward people who already experience discrimination and health disparities.

Clinical Trial of HIV Vaccine Begins in United States and South Africa
September 20, 2023
A trial of a preventive HIV vaccine candidate has begun enrollment in the United States and South Africa. The Phase 1 trial will evaluate a novel vaccine known as VIR-1388 for its safety and ability to induce an HIV-specific immune response in people.
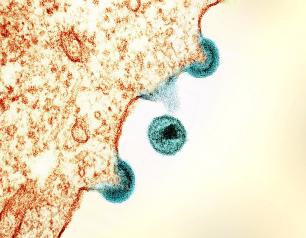
Daily Statin Reduces Heart Disease Risk Among Adults Living with HIV
July 24, 2023
A National Institutes of Health-supported study found that statins, a class of cholesterol-lowering medications, may offset the high risk of cardiovascular disease in people living with HIV by more than a third, potentially preventing one in five major cardiovascular events or premature deaths in this population. People living with HIV can have a 50-100% increased risk for cardiovascular disease.
Daily Statin Reduces the Risk of Cardiovascular Disease in People Living with HIV, Large NIH Study Finds
April 11, 2023
A National Institutes of Health (NIH) clinical trial was stopped early because a daily statin medication was found to reduce the increased risk of cardiovascular disease among people living with HIV in the first large-scale clinical study to test a primary cardiovascular prevention strategy in this population.
Experimental HIV Vaccine Regimen Safe but Ineffective, NIH Study Finds
January 18, 2023
An investigational HIV vaccine regimen tested among men who have sex with men (MSM) and transgender people was safe but did not provide protection against HIV acquisition, an independent data and safety monitoring board (DSMB) has determined. The HPX3002/HVTN 706, or “Mosaico,” Phase 3 clinical trial began in 2019 and involved 3,900 volunteers ages 18 to 60 years in Europe, North America and South America.
NIH Statement on World AIDS Day
December 1, 2022
In the 34 years since the first observance of World AIDS Day, transformational progress has been made in the global fight against HIV/AIDS, yet challenges remain. Today, we at the National Institutes of Health reflect on the 40 million lives lost to the disease and renew our commitment to the research necessary to end the global pandemic.

Early HIV diagnosis and treatment important for better long-term health outcomes
October 21, 2022
Starting antiretroviral treatment (ART) early in the course of HIV infection when the immune system is stronger results in better long-term health outcomes compared with delaying ART, according to findings presented today at the IDWeek Conference in Washington, D.C.


