37 Results
NIH Statement on World AIDS Day
December 1, 2021
Since 1988, World AIDS Day has been an annual call to end the HIV/AIDS pandemic as we remember the many who lost their lives to the disease. Considerable progress has been made since the first World AIDS Day; however, far too many people continue to acquire HIV and die from its related illnesses.
NIH Awards More than $20 Million to International HIV Database Centers
July 22, 2021
The National Institutes of Health has renewed grants to seven regional centers that compose the International epidemiology Databases to Evaluate AIDS (IeDEA), awarding $20.8 million in first-year funding. The 15-year-old IeDEA program efficiently advances knowledge about HIV by pooling and analyzing de-identified health data from more than two million people with HIV on five continents to answer research questions that individual studies cannot address. The grants are expected to last five years and to total an estimated $100 million.

Too Many People with HIV Fail to Achieve Durable Viral Suppression
November 29, 2021
Among people with HIV worldwide who are receiving antiretroviral therapy (ART), adults are getting closer to the global target of 95% achieving viral suppression, but progress among children and adolescents is lagging and long-term viral suppression among all groups remains a challenge.

To End HIV Epidemic, We Must Address Health Disparities
February 19, 2021
NIH reports that scientific strides in HIV treatment and prevention have reduced transmissions and HIV-related deaths significantly in the US.
NIH Makes Substantial New Investment in HIV Cure Research
August 17, 2021
The National Institutes of Health has awarded approximately $53 million in annual funding over the next five years to 10 research organizations in a continued effort to find a cure for HIV.
NIH Experts Call for Accelerated Research to Address Concurrent HIV and COVID-19 Pandemics
April 8, 2021
The COVID-19 pandemic is affecting people with or at risk for HIV both indirectly, by interfering with HIV treatment and prevention services, and directly, by threatening individual health. An effective response to these dual pandemics requires unprecedented collaboration to accelerate basic and clinical research, as well as implementation science to expeditiously introduce evidence-based strategies into real-world settings. This message comes from a review article co-authored by Anthony S.
NIH Researchers Identify How Two People Controlled HIV After Stopping Treatment
October 28, 2021
Research led by scientists at the National Institutes of Health has identified two distinct ways that people with HIV can control the virus for an extended period after stopping antiretroviral therapy (ART) under medical supervision. This information could inform efforts to develop new tools to help people with HIV put the virus into remission without taking lifelong medication, which can have long-term side-effects.
Antibody Infusions Prevent Acquisition of Some HIV Strains, NIH Studies Find
January 26, 2021
NIH finds that an investigational anti-HIV antibody prevented acquisition of some HIV strains, but did not significantly reduce overall acquisition.
NIH Scientists Identify Nutrient that Helps Prevent Bacterial Infection
January 15, 2021
NIAID scientists studying natural defenses against bacterial infection found that taurine helps the gut recall prior infections and kill invading bacteria.
Statement—Large Clinical Trial Will Test Combination Monoclonal Antibody Therapy for Mild/Moderate COVID-19
January 5, 2021
A NIAID-supported clinical trial has begun to evaluate a combination investigational monoclonal antibody therapy for people with mild to moderate COVID-19.
Statement—Four Potential COVID-19 Therapeutics Enter Phase 2/3 Testing in NIH ACTIV-2 Trial
February 12, 2021
Enrollment has begun to test additional investigational drugs in the NIH Accelerating COVID-19 Therapeutic Interventions and Vaccines (ACTIV) program.
Statement—Large NIH Clinical Trial Will Test Polyclonal Antibody Therapeutic for COVID-19
April 21, 2021
A Phase 2/3 trial to evaluate a new fully-human polyclonal antibody therapeutic targeted to SARS-CoV-2, called SAB-185, has begun enrolling non-hospitalized people with mild or moderate cases of COVID-19. The trial, ACTIV-2, is sponsored by the National Institute of Allergy and Infectious Diseases (NIAID), part of the National Institutes of Health. The therapeutic was developed by SAB Biotherapeutics, Inc. (Sioux Falls, South Dakota).
Experimental mRNA HIV Vaccine Safe, Shows Promise in Animals
December 9, 2021
An experimental HIV vaccine based on mRNA—the same platform technology used in two highly effective COVID-19 vaccines—shows promise in mice and non-human primates, according to scientists at the National Institute of Allergy and Infectious Diseases (NIAID), part of the National Institutes of Health.
Statement—Janssen Investigational COVID-19 Vaccine—Interim Analysis
January 29, 2021
NIH reports that an investigational COVID-19 vaccine by Janssen Pharmaceuticals appears to be safe and effective at preventing COVID-19 in adults.
HIV Vaccine Candidate Does Not Sufficiently Protect Women Against HIV Infection
August 31, 2021
An investigational HIV vaccine tested in the “Imbokodo” clinical trial conducted in sub-Saharan Africa posed no safety concerns but did not provide sufficient protection against HIV infection, according to a primary analysis of the study data. The Phase 2b proof-of-concept study, which began in November 2017, enrolled 2,637 women ages 18 to 35 years from five countries.
NIH Experts Discuss SARS-CoV-2 Viral Variants
February 12, 2021
The rise of several significant variants of SARS-CoV-2 has attracted the attention of health and science experts worldwide, NIH reports.
NIH Establishes New Childhood Asthma Clinical Research Network
April 23, 2021
The National Institute of Allergy and Infectious Diseases (NIAID), part of the National Institutes of Health, has awarded $10 million in first-year funding to establish a clinical research network called Childhood Asthma in Urban Settings (CAUSE). This nationwide network will conduct observational studies and clinical trials to improve understanding of asthma and develop treatment and prevention approaches tailored to children of low-income families living in urban communities. NIAID intends to provide approximately $70 million over seven years to support the CAUSE network.
NIH Scientists Use Human Cerebral Organoid to Test Drug for Deadly Brain Disease
March 9, 2021
After establishing a human cerebral organoid system to study CJD, NIAID researchers have developed the model to screen drugs for potential CJD treatment.
NIH Clinical Trial Evaluating Moderna COVID-19 Variant Vaccine Begins
March 31, 2021
An investigational vaccine designed to protect against the B.1.351 SARS-CoV-2 variant has been administered as part of a new Phase 1 clinical trial evaluating the vaccine candidate’s safety and immunogenicity in adult volunteers. The vaccine, known as mRNA-1273.351, was developed by the biotechnology company ModernaTX, Inc., based in Cambridge, Massachusetts. The trial is led and funded by NIAID. The trial will enroll approximately 210 healthy adult volunteers at four clinical research sites in the United States that are part of the NIAID-funded Infectious Diseases Clinical Research Consortium (IDCRC).
Intranasal Influenza Vaccine Spurs Strong Immune Response in Phase 1 Study
February 3, 2021
An experimental influenza vaccine was safe and produced a durable immune response when tested in a Phase 1 study, NIH reports.
NIH Hamster Study Evaluates Airborne and Fomite Transmission of SARS-CoV-2
August 17, 2021
National Institutes of Health scientists studying SARS-CoV-2, the virus that causes COVID-19, have defined in Syrian hamsters how different routes of virus exposure are linked to disease severity. Their study, published in Nature Communications, details the efficiency of airborne transmission between hamsters and examines how the virus replicates and causes disease throughout the respiratory system. Their work also shows that virus transmission via fomites—exposure from contaminated surface contact—is markedly less efficient than airborne transmission but does occur.
Media Availability—COVID Vaccine Booster Increases Antibody Responses, Is Protective in Rhesus Macaques
October 21, 2021
A booster dose of the mRNA-1273 COVID-19 vaccine given to rhesus macaques about six months after their primary vaccine series significantly increased levels of neutralizing antibodies against all known SARS-CoV-2 variants of concern, according to a new study from National Institutes of Health scientists and colleagues.

Lung Autopsies of COVID-19 Patients Reveal Treatment Clues
November 17, 2021
Lung autopsy and plasma samples from people who died of COVID-19 have provided a clearer picture of how the SARS-CoV-2 virus spreads and damages lung tissue.
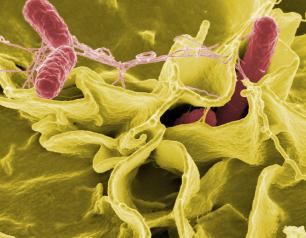
NIH Scientists Study Salmonella Swimming Behavior as Clues to Infection
January 13, 2021
NIH scientists believe they have identified a protein, that allows the Salmonella bacteria to swim straight when they are ready to infect cells.
NIAID Scientists Find a Key to Hepatitis C Entry into Cells
September 21, 2021
Understanding Structure of HCV Proteins Could Aid in Vaccine Development