15 Results
NIH Statement on World AIDS Day
December 1, 2022
In the 34 years since the first observance of World AIDS Day, transformational progress has been made in the global fight against HIV/AIDS, yet challenges remain. Today, we at the National Institutes of Health reflect on the 40 million lives lost to the disease and renew our commitment to the research necessary to end the global pandemic.

NIH Launches Clinical Trial of Three mRNA HIV Vaccines
March 14, 2022
The National Institute of Allergy and Infectious Diseases (NIAID), part of the National Institutes of Health, has launched a Phase 1 clinical trial evaluating three experimental HIV vaccines based on a messenger RNA (mRNA) platform—a technology used in several approved COVID-19 vaccines. NIAID is sponsoring the study, called HVTN 302, and the NIAID-funded HIV Vaccine Trials Network (HVTN), based at Fred Hutchinson Cancer Research Center in Seattle, is conducting the trial.
Researchers Document Third Known Case of HIV Remission Involving Stem Cell Transplant
February 15, 2022
A woman with HIV who received a cord blood stem cell transplant to treat acute myeloid leukemia has had no detectable levels of HIV for 14 months despite cessation of antiretroviral therapy (ART), according to a presentation at today’s Conference on Retroviruses and Opportunistic Infections (CROI).
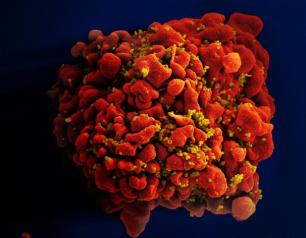
NIAID Pandemic Preparedness Plan Targets ‘Prototype’ and Priority Pathogens
February 2, 2022
The National Institute of Allergy and Infectious Diseases is focusing on preparing for a range of other viral threats that could cause a public health emergency, and according to NIAID’s new Pandemic Preparedness Plan, the institute will direct its preparedness efforts on two fronts.
Dr. Fauci Reflects on the Perpetual Challenge of Infectious Diseases
November 28, 2022
Dr. Fauci, who since 1984 has directed the National Institute of Allergy and Infectious Diseases (NIAID), part of the National Institutes of Health, reflects on his career responding to infectious disease threats.

NIH Experts Review Monkeypox Challenges
August 24, 2022
National Institutes of Health experts write that lessons learned from the public health responses to the HIV and COVID-19 pandemics should help guide the response to the current outbreak of monkeypox.
Statement by Anthony S. Fauci, M.D.
August 22, 2022
I am announcing today that I will be stepping down from the positions of Director of the National Institute of Allergy and Infectious Diseases (NIAID) and Chief of the NIAID Laboratory of Immunoregulation, as well as the position of Chief Medical Advisor to President Joe Biden. I will be leaving these positions in December of this year to pursue the next chapter of my career.
U.S. Clinical Trial Evaluating Antiviral for Monkeypox Begins
September 9, 2022
A Phase 3 clinical trial evaluating the antiviral tecovirimat, also known as TPOXX, is now enrolling adults and children with monkeypox infection in the United States. Study investigators aim to enroll more than 500 people from clinical research sites nationwide. Interested volunteers can visit the ACTG website (clinical trial A5418) for more information. The NIAID-funded Advancing Clinical Therapeutics Globally for HIV/AIDS and Other Infections (ACTG) is leading the study, which may later expand to international sites. The Eunice Kennedy Shriver National Institute of Child Health and Human Development (NICHD) at NIH is supporting several sites, including through the International Maternal Pediatric Adolescent AIDS Clinical Trials Network (IMPAACT).
Monkeypox Treatment Trial Begins in the Democratic Republic of the Congo
October 12, 2022
A clinical trial to evaluate the antiviral drug tecovirimat, also known as TPOXX, in adults and children with monkeypox has begun in the Democratic Republic of the Congo (DRC). The trial will evaluate the drug’s safety and its ability to mitigate monkeypox symptoms and prevent serious outcomes, including death. NIAID and the DRC’s National Institute for Biomedical Research (INRB) are co-leading the trial as part of the government-to-government PALM partnership. Collaborating institutions include the U.S. Centers for Disease Control and Prevention (CDC), the Institute of Tropical Medicine Antwerp, the aid organization Alliance for International Medical Action (ALIMA) and the World Health Organization (WHO).
World TB Day 2022—Invest to End TB. Save Lives
March 24, 2022
Today marks the 140th anniversary of the announcement by Dr. Robert Koch that tuberculosis (TB) is caused by the bacterium Mycobacterium tuberculosis. World TB Day is a reminder that this ancient disease remains a relentless killer. The National Institute of Allergy and Infectious Diseases (NIAID), part of the National Institutes of Health, affirms its commitment to the 2022 World TB Day theme, Invest to End TB. Save Lives, by supporting and conducting wide-ranging research aimed at reducing the health and economic impacts of TB.
Early HIV diagnosis and treatment important for better long-term health outcomes
October 21, 2022
Starting antiretroviral treatment (ART) early in the course of HIV infection when the immune system is stronger results in better long-term health outcomes compared with delaying ART, according to findings presented today at the IDWeek Conference in Washington, D.C.

Three-dose hepatitis B vaccine regimen protects people with HIV
October 20, 2022
A three-dose course of the hepatitis B vaccine HEPLISAV-B fully protected adults living with HIV who had never been vaccinated against or infected with the hepatitis B virus (HBV), according to study findings presented today at the IDWeek conference in Washington, D.C. The National Institute of Allergy and Infectious Diseases (NIAID), part of the National Institutes of Health, sponsors the ongoing Phase 3 ACTG A5379 clinical study.
New Insights into HIV Latent Cells Yield Potential Cure Targets
July 27, 2022
Scientists with the National Institute of Allergy and Infectious Diseases’ (NIAID) Vaccine Research Center (VRC) and their collaborators described how their use of cutting-edge technology revealed new insights into cellular reservoirs of HIV and what those observations could mean for the next steps in HIV cure research.
Combination Anti-HIV Antibody Infusions Suppress Virus for Prolonged Period
June 1, 2022
According to a small study published today in the journal Nature, individuals with HIV who began taking antiretroviral therapy (ART) in the early stages of infection achieved a lengthy period of HIV suppression without ART after receiving two broadly neutralizing anti-HIV antibodies (bNAbs).
Leadership Transition at the NIAID Vaccine Research Center
February 16, 2022
Dr. Fauci expresses gratitude to John R. Mascola, M.D., as he announces his retirement as Director of the Dale and Betty Bumpers Vaccine Research Center at the National Institute of Allergy and Infectious Diseases.

