57 Results
NIH Statement on World AIDS Day
December 1, 2022
In the 34 years since the first observance of World AIDS Day, transformational progress has been made in the global fight against HIV/AIDS, yet challenges remain. Today, we at the National Institutes of Health reflect on the 40 million lives lost to the disease and renew our commitment to the research necessary to end the global pandemic.

Daily Statin Reduces Heart Disease Risk Among Adults Living with HIV
July 24, 2023
A National Institutes of Health-supported study found that statins, a class of cholesterol-lowering medications, may offset the high risk of cardiovascular disease in people living with HIV by more than a third, potentially preventing one in five major cardiovascular events or premature deaths in this population. People living with HIV can have a 50-100% increased risk for cardiovascular disease.
NIH Statement on World AIDS Day
December 1, 2021
Since 1988, World AIDS Day has been an annual call to end the HIV/AIDS pandemic as we remember the many who lost their lives to the disease. Considerable progress has been made since the first World AIDS Day; however, far too many people continue to acquire HIV and die from its related illnesses.
NIH Makes Substantial New Investment in HIV Cure Research
August 17, 2021
The National Institutes of Health has awarded approximately $53 million in annual funding over the next five years to 10 research organizations in a continued effort to find a cure for HIV.
World AIDS Day 2023
December 1, 2023
On this 35th World AIDS Day, the National Institutes of Health (NIH) joins its partners in honoring the lives lost due to the HIV pandemic. For decades, this virus has exacted a tragic toll, affecting people, families, and communities worldwide, threatening social and economic development, and exacerbating stigma, often toward people who already experience discrimination and health disparities.

NIH Clinical Trial of Tuberculous Meningitis Drug Regimen Begins
December 7, 2023
A trial of a new drug regimen to treat tuberculous meningitis (TBM) has started enrolling adults and adolescents in several countries where tuberculosis (TB) is prevalent. The trial will include 330 participants aged 15 years and older who have or are likely to have TBM based on signs and symptoms, including people living with and without HIV. Because pregnant women are eligible to enroll in this study with appropriate consent, a small number of pregnant women are expected to be included.
NIH Launches Clinical Trial of Three mRNA HIV Vaccines
March 14, 2022
The National Institute of Allergy and Infectious Diseases (NIAID), part of the National Institutes of Health, has launched a Phase 1 clinical trial evaluating three experimental HIV vaccines based on a messenger RNA (mRNA) platform—a technology used in several approved COVID-19 vaccines. NIAID is sponsoring the study, called HVTN 302, and the NIAID-funded HIV Vaccine Trials Network (HVTN), based at Fred Hutchinson Cancer Research Center in Seattle, is conducting the trial.
Novel Vaccine Concept Generates Immune Responses that Could Produce Multiple Types of HIV Broadly Neutralizing Antibodies
May 30, 2024
Using a combination of cutting-edge immunologic technologies, researchers have successfully stimulated animals’ immune systems to induce rare precursor B cells of a class of HIV broadly neutralizing antibodies (bNAbs). The findings, published today in Nature Immunology, are an encouraging, incremental step in developing a preventive HIV vaccine.
NIAID Selects Sarah Read as Principal Deputy Director
October 2, 2024
The National Institute of Allergy and Infectious Diseases (NIAID), part of the National Institutes of Health (NIH), has named Sarah W. Read, M.D., MHS, as the principal deputy director for the institute.

NIH Experts Call for Accelerated Research to Address Concurrent HIV and COVID-19 Pandemics
April 8, 2021
The COVID-19 pandemic is affecting people with or at risk for HIV both indirectly, by interfering with HIV treatment and prevention services, and directly, by threatening individual health. An effective response to these dual pandemics requires unprecedented collaboration to accelerate basic and clinical research, as well as implementation science to expeditiously introduce evidence-based strategies into real-world settings. This message comes from a review article co-authored by Anthony S.
Researchers Document Third Known Case of HIV Remission Involving Stem Cell Transplant
February 15, 2022
A woman with HIV who received a cord blood stem cell transplant to treat acute myeloid leukemia has had no detectable levels of HIV for 14 months despite cessation of antiretroviral therapy (ART), according to a presentation at today’s Conference on Retroviruses and Opportunistic Infections (CROI).
Too Many People with HIV Fail to Achieve Durable Viral Suppression
November 29, 2021
Among people with HIV worldwide who are receiving antiretroviral therapy (ART), adults are getting closer to the global target of 95% achieving viral suppression, but progress among children and adolescents is lagging and long-term viral suppression among all groups remains a challenge.

Daily Statin Reduces the Risk of Cardiovascular Disease in People Living with HIV, Large NIH Study Finds
April 11, 2023
A National Institutes of Health (NIH) clinical trial was stopped early because a daily statin medication was found to reduce the increased risk of cardiovascular disease among people living with HIV in the first large-scale clinical study to test a primary cardiovascular prevention strategy in this population.
Tools Underestimate Cardiovascular Event Risk in People with HIV
March 4, 2024
The elevated cardiovascular disease risk among people with HIV is even greater than predicted by a standard risk calculator in several groups, including Black people and cisgender women, according to analyses from a large international clinical trial primarily funded by the National institutes of Health and presented at the 2024 Conference on Retroviruses and Opportunistic Infections (CROI) in Denver.
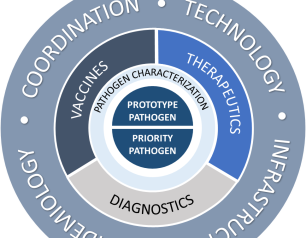
NIAID Pandemic Preparedness Plan Targets ‘Prototype’ and Priority Pathogens
February 2, 2022
The National Institute of Allergy and Infectious Diseases is focusing on preparing for a range of other viral threats that could cause a public health emergency, and according to NIAID’s new Pandemic Preparedness Plan, the institute will direct its preparedness efforts on two fronts.
To End HIV Epidemic, We Must Address Health Disparities
February 19, 2021
NIH reports that scientific strides in HIV treatment and prevention have reduced transmissions and HIV-related deaths significantly in the US.
NIH Statement on World AIDS Day
December 2, 2024
Together with our partners, the National Institutes of Health (NIH) commemorates World AIDS Day and affirms our commitment to bolstering the extraordinary gains achieved in HIV science and to persevering until we end HIV-related illness and stigma. As we mark this observance, we celebrate the people who enable scientific progress, honor the loved ones and leaders we have lost, and reflect on the work that remains to ensure the health and life quality of all people affected by, and living with, HIV.
Dr. Fauci Reflects on the Perpetual Challenge of Infectious Diseases
November 28, 2022
Dr. Fauci, who since 1984 has directed the National Institute of Allergy and Infectious Diseases (NIAID), part of the National Institutes of Health, reflects on his career responding to infectious disease threats.

NIH Experts Review Monkeypox Challenges
August 24, 2022
National Institutes of Health experts write that lessons learned from the public health responses to the HIV and COVID-19 pandemics should help guide the response to the current outbreak of monkeypox.
Statement by Anthony S. Fauci, M.D.
August 22, 2022
I am announcing today that I will be stepping down from the positions of Director of the National Institute of Allergy and Infectious Diseases (NIAID) and Chief of the NIAID Laboratory of Immunoregulation, as well as the position of Chief Medical Advisor to President Joe Biden. I will be leaving these positions in December of this year to pursue the next chapter of my career.
Investigational Three-Month TB Regimen Is Safe but Ineffective, NIH Study Finds
July 5, 2023
The first clinical trial of a three-month tuberculosis (TB) treatment regimen is closing enrollment because of a high rate of unfavorable outcomes with the investigational course of treatment. Advancing Clinical Therapeutics Globally for HIV/AIDS and Other Infections (ACTG) 5362, also known as the CLO-FAST trial, sought to evaluate the safety and efficacy of a three-month clofazimine- and high-dose rifapentine-containing regimen. An interim data analysis showed that participants taking the investigational regimen experienced ongoing or recurring TB at rates above thresholds set in the study protocol.
NIAID Appoints Ted Pierson as New Vaccine Research Center Director
April 25, 2023
The National Institute of Allergy and Infectious Diseases (NIAID), part of the National Institutes of Health, has named Theodore (Ted) C. Pierson, Ph.D., as the new director of its Dale and Betty Bumpers Vaccine Research Center (VRC) in Bethesda, MD.

Statement—Large Clinical Trial Will Test Combination Monoclonal Antibody Therapy for Mild/Moderate COVID-19
January 5, 2021
A NIAID-supported clinical trial has begun to evaluate a combination investigational monoclonal antibody therapy for people with mild to moderate COVID-19.
Statement—Four Potential COVID-19 Therapeutics Enter Phase 2/3 Testing in NIH ACTIV-2 Trial
February 12, 2021
Enrollment has begun to test additional investigational drugs in the NIH Accelerating COVID-19 Therapeutic Interventions and Vaccines (ACTIV) program.
Statement—Large NIH Clinical Trial Will Test Polyclonal Antibody Therapeutic for COVID-19
April 21, 2021
A Phase 2/3 trial to evaluate a new fully-human polyclonal antibody therapeutic targeted to SARS-CoV-2, called SAB-185, has begun enrolling non-hospitalized people with mild or moderate cases of COVID-19. The trial, ACTIV-2, is sponsored by the National Institute of Allergy and Infectious Diseases (NIAID), part of the National Institutes of Health. The therapeutic was developed by SAB Biotherapeutics, Inc. (Sioux Falls, South Dakota).