
Dr. Michael Rybak, director of the Anti-Infective Research Laboratory, Wayne State University
Since the early 1940s, antibiotics have been relied upon to effectively treat a range of infections from pneumonia to sexually transmitted diseases. While these drugs have saved millions of lives, their effectiveness is waning for several reasons, including inappropriate prescribing practices by health care providers and misuse by patients, such as not taking the medicines as prescribed. This growing public health crisis of antibiotic resistance is made worse by the fact that few new antibiotic medicines are immediately on the horizon. The challenge for society is determining how to ensure that currently available antibiotics remain effective in the future while scientists work to find new treatments.
NIAID grantee Michael Rybak, Pharm.D., M.P.H., and his team at Wayne State University in Detroit, Michigan, are conducting laboratory-based research to determine how best to preserve existing antibiotics and reveal whether certain bacteria are becoming resistant to specific medications. In the laboratory, Dr. Rybak’s group studies the characteristics of antibiotics using unique devices called in vitro pharmacokinetic and pharmacodynamic (PK/PD) models. These models are able to simulate the administration of antibiotics in humans to treat a variety of bacterial infections. First, the models are filled with the bacterial strain of interest and then specific media used to support the growth of the bacteria are pushed through a series of tubes and computerized pumps. Antibiotics are then administered in a way that mimics the drug concentrations that would be used to treat a patient for a specific bacterial infection.
Using the models, Dr. Rybak’s team can evaluate how effective the antibiotic is at killing the bacteria as well as whether the targeted bacteria become resistant to the treatment. By manipulating the amount of antibiotic and how often it is administered, the researchers can help establish the optimal dose and frequency needed to stop the infection while preventing the emergence of drug resistance.
Matching Lab Results to Patients
Laboratory testing is an efficient and safe way to study antibiotics and dosages, but it is also important to study actual patient outcomes. In partnership with several colleagues, Dr. Rybak studies hospital patient charts to determine how patients with various types of bacterial infections respond to antibiotics. Combining this patient information with laboratory data, he has been able to help physicians choose the appropriate drug regimens to treat very challenging bacterial infections.
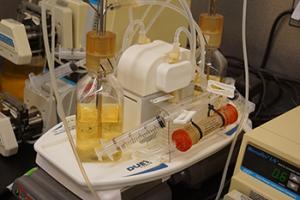
Pharmacokinetic and pharmacodynamic (PK/PD) models used by Dr. Rybak’s team to simulate antibiotic use in humans.
In one recent case, Dr. Rybak and his team helped a patient with a serious, multidrug-resistant Staphylococcus aureus (MRSA) bloodstream infection. Specifically, Dr. Rybak and his team found in the laboratory setting that a high dose of the antibiotic daptomycin combined with the antibiotics trimethoprim and sulfamethoxazole were highly effective against MRSA. Based on this finding, Dr. Rybak’s group recommended this antibiotic combination to the physicians treating the MRSA patient, and it worked—the patient’s MRSA infection was successfully cured. Since that time, Dr. Rybak has also recommended other successful treatments for hospital-based patients with bacterial infections.
Looking Ahead
Dr. Rybak’s goal is to continue to be able to predict when and how drug resistance will develop in response to antibiotic exposure and to provide that information to health care providers, so that optimal medications and dosages can be determined.
“Antibiotic resistance has become a real public health problem,” said Dr. Rybak. “The ability to best match a drug to a specific infection could potentially reduce hospital stays, decrease healthcare costs, diminish the risk for antibiotic resistance, and save lives.”
References
Rybak MJ, Vidaillac C, Steed ME. Novel Daptomycin Combinations against Daptomycin-Nonsusceptible Methicillin-Resistant Staphylococcus aureus in an In Vitro Model of Simulated Endocardial Vegetations. Antimicrobial Agents and Chemotherapy. 54(12): 5187-5192 (2010).
Rybak MJ, Hasan M, Woodruff AE, Steed ME, Avery LM. Daptomycin-Nonsusceptible Vancomycin-Intermediate Staphylococcus aureus Vertebral Osteomyelitis Complicated by Bacteremia Treated with High-Dose Daptomycin and Trimethoprim-Sulfamethoxazole. Antimicrobial Agents and Chemotherapy 56(11): 5590-5993 (2012).

