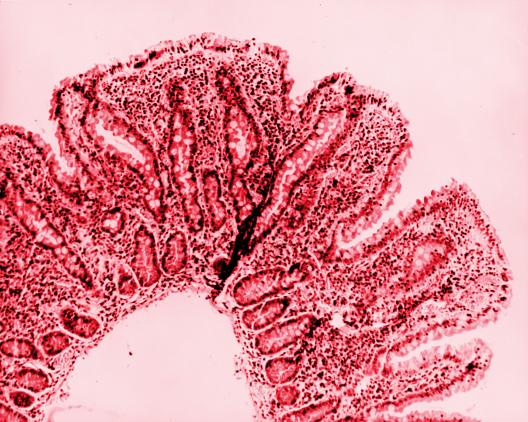
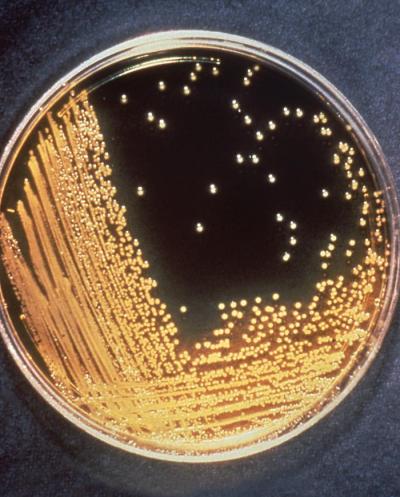
The Early Phase Clinical Trial Units (EPCTUs) support the design, development, implementation, and conduct of Phase 0 to Phase 2 clinical trials, including proof-of-concept studies in healthy, special, and disease-specific populations, carried out in an expeditious and efficient manner, aided by bioanalysis when necessary, against viral, bacterial, parasitic, and fungal pathogens and emerging and re-emerging infectious diseases. The candidate products assessed through the EPCTUs may include licensed or investigational products such as small molecules, monoclonal antibodies, i
Research
Research Areas
Grants & Contracts
Clinical Trials
News & Events
About NIAID
Website Policies and Notices