The HIV/AIDS clinical trials units (CTUs) provide scientific and administrative expertise, as well as the infrastructure to conduct clinical trials within the HIV/AIDS clinical trials networks. Each CTU supports up to eight clinical research sites.
Table of Contents
List of HIV/AIDS Clinical Trials Units
In November 2020, NIAID awarded grants to 35 U.S. and international institutions selected as HIV/AIDS CTUs. View information about these awards, listed alphabetically by institution, in the table below:
| Grant Number | Title | Principal Investigator(s) | Institution |
|---|---|---|---|
| 2 UM1 AI069438-15 | PERU Clinical Trial Unit | Jorge L. Sanchez, Alberto La Rosa | Asociacion Civil Impacta Salud y Educacion |
| 1 UM1 AI154463-01 | Aurum South and East African Clinical Trials Unit | Gavin J. Churchyard | Aurum Institute NPC |
| 2 UM1 AI069412-15 | Boston/Jackson/Providence HIV Clinical Trials Unit (BJP HIV CTU) | Daniel R. Kuritzkes, Lindsey R. Baden, Kenneth H. Mayer | Brigham and Women's Hospital |
| 2 UM1 AI069501-15 | Case Clinical Trials Unit | Benigno Rodriguez | Case Western Reserve University |
| 2 UM1 AI069469-15 | KwaZulu-Natal Clinical Trials Unit | Quarraisha Abdool Karim | Centre for the AIDS Programme of Research in South Africa |
| 2 UM1 AI069399-15 | Thailand HIV/AIDS and Infectious Disease Clinical Trials Unit (THAI CTU) | Khuanchai Supparatpinyo, Kiat Ruxrungtham | Chiang Mai University |
| 2 UM1 AI069470-15 | Columbia Collaborative HIV/AIDS Clinical Trials Unit | Magdalena E. Sobieszczyk | Columbia University Health Sciences |
| 1 UM1 AI154468-01 | ICAP Clinical Trials Unit | Wafaa M. El-Sadr, Jessica E. Justman | Columbia University Health Sciences |
| 2 UM1 AI069418-15 | Emory-CDC CTU | Jeffrey Lennox, Carlos del Rio | Emory University |
| 2 UM1 AI069481-15 | Seattle-Lausanne Clinical Trials Unit | Margaret J. McElrath | Fred Hutchinson Cancer Research Center |
| 2 UM1 AI069476-14 | The FIOCRUZ Therapeutic and Prevention HIV/AIDS Clinical Trials Unit. | Beatriz Grinsztejn | Fundacao Oswaldo Cruz |
| 1 UM1 AI154466-01 | The District of Columbia Clinical Trials Unit (DC CTU) | Manya Magnus, Sarah Henn, Gary L. Simon | George Washington University |
| 2 UM1 AI069421-15 | GHESKIO Clinical Trials Unit | Jean W. Pape | GHESKIO Center |
| 2 UM1 AI069456-15 | Botswana-Harvard T.H. Chan School of Public Health AIDS Initiative Partnership CTU | Shahin Lockman, Joseph M. Makhema | Harvard School of Public Health |
| 2 UM1 AI108568-08 | The Military HIV Research Program (MHRP) Clinical Trial Unit | Merlin L. Robb, Julie Ake | Henry M. Jackson Foundation for the Advancement of Military Medicine |
| 2 UM1 AI069465-15 | Johns Hopkins University Baltimore India Clinical Trail Unit (JHUBI CTU) | Amita Gupta, Charles W. Flexner | Johns Hopkins University |
| 2 UM1 AI069530-15 | The Johns Hopkins University - Uganda Clinical Trials Unit | Mary Glenn Fowler, Philippa Musoke, Aaron A. Tobian | Johns Hopkins University |
| 2 UM1 AI069518-15 | The Johns Hopkins University-Blantyre Clinical Trials Unit | Taha E. Taha | Johns Hopkins University |
| 2 UM1 AI069471-15 | Chicago Clinical Trials Unit | Babafemi O. Taiwo | Northwestern University at Chicago |
| 2 UM1 AI069496-15 | San Francisco Bay Clinical Trials Unit (CTU) | Susan Buchbinder, Diane V. Havlir | Public Health Foundation Enterprises |
| 2 UM1 AI069521-15 | Stellenbosch University Clinical Trials Unit | Mark F. Cotton, Andreas Diacon, Anneke C. Hesseling | Stellenbosch University Tygerberg Campus |
| 2 UM1 AI069452-15 | A-Z Clinical Trials Unit | Edgar T. Overton | University of Alabama at Birmingham |
| 2 UM1 AI069424-15 | UCLA AIDS Prevention and Treatment Clinical Trials Unit | Judith S. Currier, Raphael J. Landovitz | University of California, Los Angeles |
| 2 UM1 AI069536-15 | HIV Centers for Underrepresented Populations in Research Clinical Trials Unit (CURE CTU) | Stephen A. Spector | University of California, San Diego |
| 2 UM1 AI069432-15 | UCSD Collaborative Clinical Trials Unit | Constance A. Benson | University of California, San Diego |
| 2 UM1 AI069436-15 | UZ-UCSF CTU | Zvavahera (Mike) Chirenje | University of California, San Francisco |
| 2 UM1 AI069519-15 | HIV/AIDS Clinical Trials Units University of Cape Town CTU 2020 to 2027 | Linda-Gail Bekker, Catherine Orrell | University of Cape Town |
| 2 UM1 AI069423-15 | University of North Carolina Global HIV Prevention and Treatment Clinical Trials Unit | Joseph J. Eron, Mina C. Hosseinipour, David A. Wohl | University of North Carolina Chapel Hill |
| 2 UM1 AI069534-15 | University of Pennsylvania HIV Clinical Trials Unit | Ian Frank, Pablo Tebas | University of Pennsylvania |
| 2 UM1 AI069494-15 | Pitt-Ohio State Clinical Trials Unit | John W. Mellors, Susan L. Koletar, Sharon Riddler | University of Pittsburgh at Pittsburgh |
| 2 UM1 AI069511-15 | University of Rochester HIV/AIDS Clinical Trials Unit | Michael C. Keefer, Stephen Dewhurst | University of Rochester |
| 2 UM1 AI069439-16 | Vanderbilt HIV Clinical Trials Unit (CTU) | David W. Haas, Spyros A. Kalams, Rachel M. Presti | Vanderbilt University Medical Center |
| 2 UM1 AI069419-15 | Weill Cornell Medicine - Rutgers New Jersey Medical School Clinical Trials Unit | Roy M. Gulick | Weill Medical College of Cornell University |
| 2 UM1 AI069453-15 | PHRU-Setshaba Clinical Trials Unit | Glenda E. Gray, Lerato Mohapi, Ravindre Panchia | Wits Health Consortium (PTY), LTD |
| 2 UM1 AI069463-15 | Wits HIV Research Group Clinical Trial Unit (CTU) | Ian M. Sanne, Helen Rees | Wits Health Consortium (PTY), LTD |
Map of HIV/AIDS Clinical Research Sites
Collectively, the 35 CTUs support 101 clinical research sites in 18 countries across North America, South America, Africa and Asia. This includes 45 sites in the United States. View a map of the sites below:
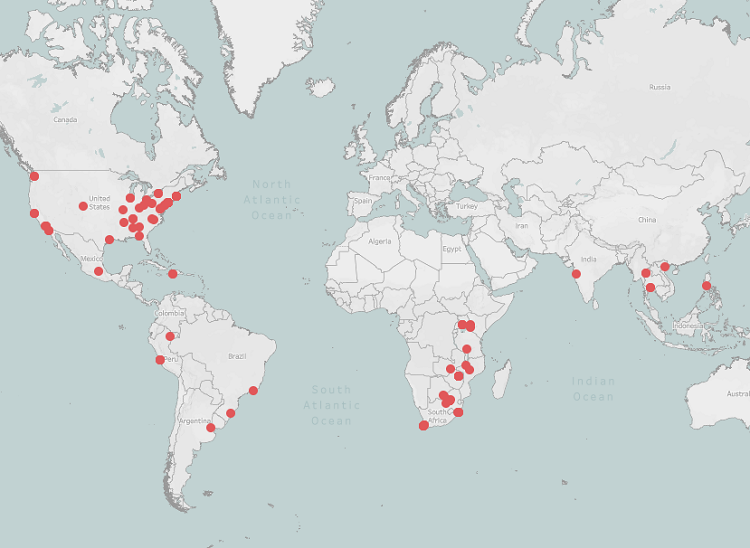
World map showing locations of the HIV/AIDS clinical research sites (CRSs). Each of the 101 CRSs is represented by a red circle. Note that some circles overlap due to proximity.

