NIAID-led scientists’ discovery of a hidden gene variant that causes some cases of a devastating inherited disease will enable earlier diagnosis of the disorder in people with the variant, facilitating earlier medical care that may prolong their lives. The researchers are working on a treatment for this unusual form of the rare autoimmune disease, known as APECED, and have traced its evolutionary origins. The findings are published in the journal Science Translational Medicine.
APECED—short for autoimmune polyendocrinopathy-candidiasis-ectodermal dystrophy—causes multi-organ dysfunction, usually beginning in childhood, and can kill up to 30% of people with the syndrome. If diagnosed early and treated by a multidisciplinary healthcare team, however, people with APECED can survive into adulthood. Scientists in NIAID’s Laboratory of Clinical Immunology and Microbiology (LCIM) have developed a world-class APECED diagnostic and treatment program, currently caring for more than 100 patients as part of an observational study and serving as a resource for clinicians across the globe.
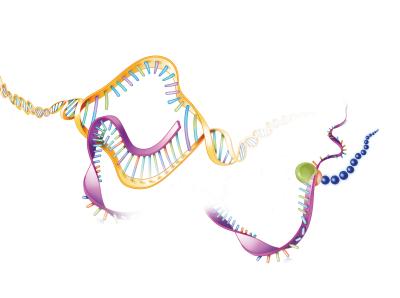
APECED is caused by mutations in a gene called AIRE, which provides instructions for making a protein that keeps the immune system’s T cells from attacking the body’s tissues and organs. These genetic mutations reduce or eliminate the protein’s normal function, leading to autoimmunity.
Most people with APECED are diagnosed based on their clinical signs and symptoms as well as on genetic testing that confirms they have a disease-causing mutation in the AIRE gene. However, as the LCIM team studied people who came to NIH with APECED, they found 17 study participants with clinical signs and symptoms of the disease but no detectable mutations in AIRE. These participants shared two notable characteristics. The families of 15 of the 17 participants were wholly or partly from Puerto Rico, a relatively small, self-contained geographic area, suggesting that the individuals’ disease might have the same genetic cause. In addition, all 17 participants had the same harmless mutation to a single building block, or nucleotide, in both copies of their AIRE gene (one inherited from each parent). This suggested they all might have a similar stretch of genetic material in or around AIRE. These clues led the researchers to start hunting for a unique genetic mechanism that could be causing APECED in the group.
The Quest for a Genetic Cause
Using technologies called whole-exome sequencing and whole-genome sequencing, the scientists determined the order of all the nucleotides in the DNA of each study participant. By examining and comparing these genetic sequences, the researchers discovered that the 17 participants had the same mutation to a single nucleotide located in a different part of the AIRE gene than the mutations commonly known to cause APECED. APECED-causing mutations usually occur in parts of the AIRE gene called “exons,” which contain the DNA code for the protein. The mutations also sometimes occur at either end of the large, non-coding sections of AIRE called “introns,” which are located in-between the exons. The newly discovered mutation was in the middle of an AIRE intron rather than at either end, so how it caused disease was initially unclear.
To solve this puzzle, the researchers examined what happens when the version of AIRE with this mid-intron mutation gets transcribed into mature messenger RNA (mRNA), the protein precursor. Normally, a molecule called a spliceosome detects the boundaries between introns and exons, cuts out the exons, and “pastes” them together in order. The scientists discovered that the mid-intron AIRE mutation fools the spliceosome into “thinking” that part of the intron is an exon, leading it to cut and paste part of the intron—extraneous genetic material—into the mature mRNA. This gives cells instructions to make an AIRE protein with an incorrect amino-acid sequence at one end. The researchers predicted and then showed that this protein can’t function normally, confirming that the mid-intron AIRE mutation causes APECED in the 17 study participants who previously lacked a genetic diagnosis.
The scientists anticipate that the newly discovered AIRE variant will be added to genetic screening panels given to people who doctors suspect have APECED or who have a family history of the disease. This could facilitate earlier diagnosis and treatment of people with the mid-intron AIRE mutation, potentially prolonging their lives. It will also enable these individuals to receive genetic counseling to inform their family planning decisions. According to the researchers, the new findings also suggest that there may be other undiscovered, mid-intron mutations that cause APECED or other inherited diseases.
A Potential Treatment in the Making
Now NIAID LCIM scientists are working on a treatment for APECED caused by the mid-intron mutation. They engineered five different strings of nucleic acids, known as antisense oligonucleotides (ASOs), designed to hide the mutation from the spliceosome. Laboratory testing in cells with the mid-intron AIRE mutation showed that one ASO worked. Unable to “see” the mutation, the spliceosome cut out the correct AIRE exons and pasted them together to make mature mRNA that could be translated into a normal AIRE protein. Next, the researchers will test this mutation-masking tool in a mouse model of APECED with this specific mid-intron mutation. They expect results in two to three years.
ASOs are an emerging form of treatment for rare genetic diseases, sometimes custom-made for just one person.
Origins of the Mutation
Through genetic and statistical analyses, the researchers estimated that the mid-intron mutation first occurred about 450 years ago. This timing coincides with when the first Europeans colonized Puerto Rico, hailing from the Cádiz province of Spain. Notably, one of the two study participants who did not have Puerto Rican ancestry also was from Cádiz and had the same set of DNA variants on one of his chromosomes as the participants with Puerto Rican ancestry. According to the researchers, these findings suggest that one or a few early Spanish colonizers of Puerto Rico carried the mid-intron AIRE mutation, and it eventually became a major cause of APECED in the Puerto Rican population. Further studies are needed to determine the prevalence of this cause of APECED among Puerto Ricans and other populations with Spanish ancestry.
By contrast, one member of the study cohort had no known Puerto Rican or Spanish ancestry and did not share the same set of DNA variants as the other 16 participants. The investigators say this suggests that the mid-intron AIRE mutation also emerged independently in North America and will likely be found in additional Americans with APECED who do not have Puerto Rican or Spanish ancestry.
Note: APECED is also known as APS-1, short for autoimmune polyglandular syndrome type 1.
References
S Ochoa et al. A deep intronic splice-altering AIRE variant causes APECED syndrome through antisense oligonucleotide-targetable pseudoexon inclusion. Science Translational Medicine DOI: 10.1126/scitranslmed.adk0845 (2024).
D Karishma et al. Antisense oligonucleotides: an emerging area in drug discovery and development. Journal of Clinical Medicine DOI: 10.3390/jcm9062004 (2020).
F Collins. One little girl’s story highlights the promise of precision medicine. NIH Director’s Blog. https://directorsblog.nih.gov/tag/milasen/ Oct. 23, 2019. Accessed Oct. 30, 2024.