Medical Virology Section
Jeffrey I. Cohen, M.D.
Chief, Laboratory of Infectious Diseases
Chief, Medical Virology Section
Provides direct clinical care to patients at NIH Clinical Center

Highlights
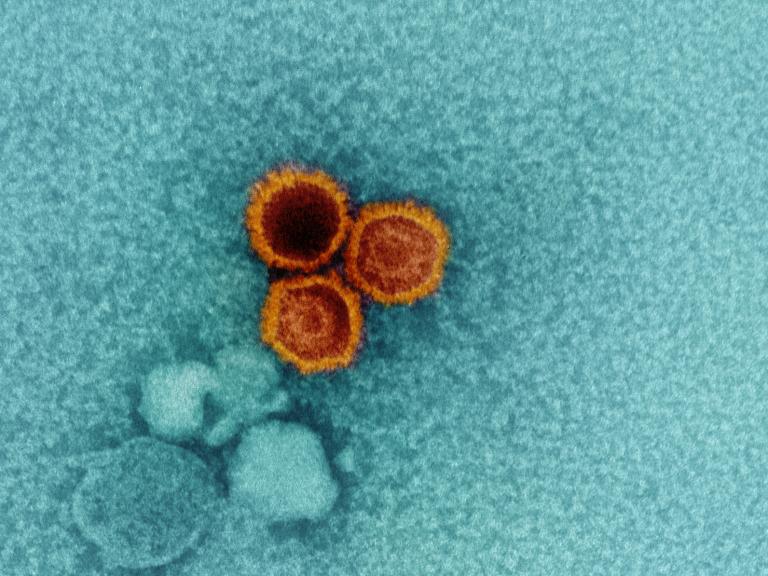
NIH Scientists Find Weak Points on Epstein-Barr Virus
March 12, 2024
Studies of interactions between two lab-generated monoclonal antibodies (mAbs) and an essential Epstein-Barr virus (EBV) protein have uncovered targets that could be exploited in designing treatments and vaccines for this extremely common virus. Study findings were published in the journal Immunity.
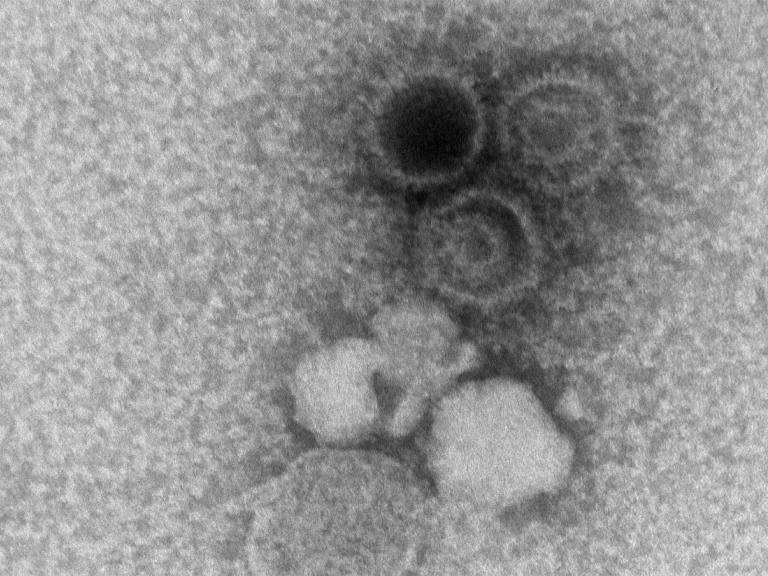
Experimental Monoclonal Antibodies Show Promise Against Epstein-Barr Virus
October 27, 2022
A panel of investigational monoclonal antibodies (mAbs) targeting different sites of the Epstein-Barr virus (EBV) blocked infection when tested in human cells in a laboratory setting. Moreover, one of the experimental mAbs provided nearly complete protection against EBV infection and lymphoma when tested in mice. The results appear online today in the journal Immunity.
Major Areas of Research
- Development of vaccines against human herpesviruses
- Development of monoclonal antibodies and other therapeutics against herpesviruses
- Identification of cellular mutations and immune defects in patients with severe herpesvirus infections
- Pathogenesis of human virus infections in vitro and in vivo
- Analysis of SARS-CoV-2-specific antibody responses
Program Description
We study the pathogenesis and clinical aspects of human herpesviruses, including Epstein-Barr virus (EBV), herpes simplex virus (HSV), varicella-zoster virus (VZV), and cytomegalovirus (CMV). The laboratory focuses on vaccine development, discovery of monoclonal antibodies to viral proteins, and identification of cellular mutations in patients with severe herpesvirus infections. Recent findings include development of candidate vaccines for HSV and EBV; identification of cellular genes that predispose to severe EBV infections; and monoclonal antibodies to EBV. Clinical projects complement the laboratory studies. These include a phase I study of an EBV nanoparticle vaccine, studies of patients with severe virus infections to define genetic variants associated with the disease, studies of patients with chronic active EBV disease.
Biography
Education
M.D., Johns Hopkins University
Dr. Cohen received his M.D. from The Johns Hopkins University and was a resident in medicine at Duke University. Following a medical staff fellowship at the National Institutes of Health (NIH), he was a clinical fellow in infectious diseases at the Brigham and Women’s Hospital and an instructor in medicine at Harvard University. He returned to NIH, where he was the chief of the Medical Virology Section in the Laboratory of Clinical Infectious Diseases until 2010. In June 2010, Dr. Cohen became chief of the Laboratory of Infectious Diseases.
Memberships
- American Association of Physicians
- American Society for Clinical Investigation
- Infectious Diseases Society of America
- American Academy of Microbiology
Editorial Boards
- Journal of Virology
- Virology
- Journal of Infectious Diseases
- Virus Genes
- Associate Editor Fields Virology
Clinical Studies
Genetic Studies of Chronic Active Epstein-Barr Disease - NCT00032513
Viral Infections in Healthy and Immunocompromised Hosts - NCT01306084
Selected Publications
Dropulic LK, Oestreich MC, Pietz HL, Laing KJ, Hunsberger S, Lumbard K, Garabedian D, Turk SP, Chen A, Hornung RL, Seshadri C, Smith MT, Hosken NA, Phogat S, Chang LJ, Koelle DM, Wang K, Cohen JI. A Randomized, Double-Blinded, Placebo-Controlled, Phase 1 Study of a Replication-Defective Herpes Simplex Virus (HSV) Type 2 Vaccine, HSV529, in Adults With or Without HSV Infection. J Infect Dis. 2019 Aug 9;220(6):990-1000.
Bu W, Joyce MG, Nguyen H, Banh DV, Aguilar F, Tariq Z, Yap ML, Tsujimura Y, Gillespie RA, Tsybovsky Y, Andrews SF, Narpala SR, McDermott AB, Rossmann MG, Yasutomi Y, Nabel GJ, Kanekiyo M, Cohen JI. Immunization with Components of the Viral Fusion Apparatus Elicits Antibodies That Neutralize Epstein-Barr Virus in B Cells and Epithelial Cells. Immunity. 2019 May 21;50(5):1305-1316.e6.
Wang K, Hoshino Y, Dowdell K, Bosch-Marce M, Myers TG, Sarmiento M, Pesnicak L, Krause PR, Cohen JI. Glutamine supplementation suppresses herpes simplex virus reactivation. J Clin Invest. 2017 Jun 30;127(7):2626-2630.
Li Q, Bu W, Gabriel E, Aguilar F, Hoshino Y, Miyadera H, Hess C, Hornung RL, Roy A, Cohen JI. HLA-DQ β1 alleles associated with Epstein-Barr virus (EBV) infectivity and EBV gp42 binding to cells. JCI Insight. 2017 Feb 23;2(4):e85687.
Sadaoka T, Depledge DP, Rajbhandari L, Venkatesan A, Breuer J, Cohen JI. In vitro system using human neurons demonstrates that varicella-zoster vaccine virus is impaired for reactivation, but not latency. Proc Natl Acad Sci U S A. 2016 Apr 26;113(17):E2403-12.
Kanekiyo M, Bu W, Joyce MG, Meng G, Whittle JR, Baxa U, Yamamoto T, Narpala S, Todd JP, Rao SS, McDermott AB, Koup RA, Rossmann MG, Mascola JR, Graham BS, Cohen JI, Nabel GJ. Rational Design of an Epstein-Barr Virus Vaccine Targeting the Receptor-Binding Site. Cell. 2015 Aug 27;162(5):1090-100.\
Research Group
Development of vaccines and monoclonal antibodies for human herpesviruses, studies of genetic variants and immune defects in persons with severe virus infections.

