The Research Technologies Branch (RTB) Biological Imaging Section (BIS) offers a wide range of advanced equipment for microscopic examination of samples. In addition to instrumentation, the facility offers advice on experimental design, instrument configuration, and optimal image collection. Information and assistance in post-collection data analysis such as quantification, colocalization, counting of objects, rates of movement, particle tracking, surface reconstruction, and segmentation, also are provided.
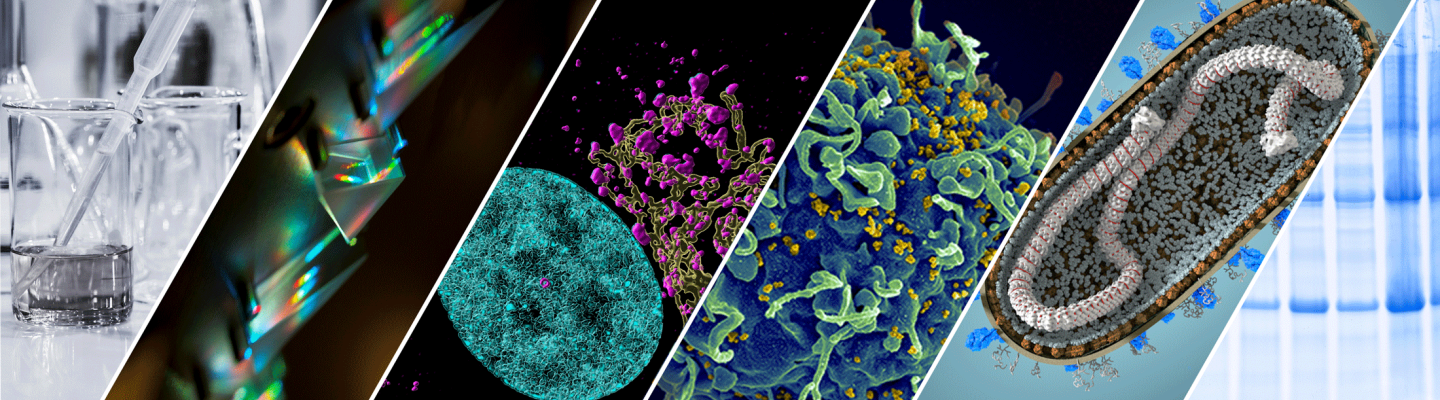
IVDN with monocytes - human
Major Areas of Support
- Confocal microscopy
- Intravital imaging
- Fluorescence lifetime imaging
- Post-collection image quantification and deconvolution
- Scientific Poster Printing
Additional Resources
- NIAID Imaris licenses
- NIAID Huygens licenses
- Image processing workstations
Instrumentation
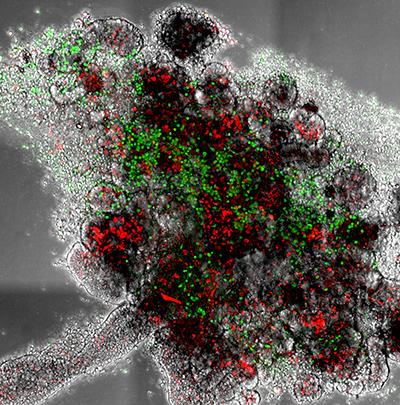
DsRed Parasites in sandfly gut
Imaging Instrumentation
- State-of-the-art confocal microscopes
- Multi-laser intravital imaging systems
- TIRF microscope
- Laser micro-capture system
- Widefield fluorescence microscopes
- Automated fluorescence stereo microscope
Preparatory Instrumentation
- Cryostats
- Vibratomes
- Surgical stereo microscopes
Advanced Intravital Microscopy
Observation of the immune response to infectious disease is important to understanding of the underlying biological processes.
The Biological Imaging Section has 4 state-of-the-art multiphoton imaging systems for intravital imaging. All three instruments are equipped with microscope enclosures for 37C incubation, as well as anesthesia for mice. Three of the instruments have dual IR lasers for excitation of probes across the IR spectrum. Additionally, the BIS is one of only a few laboratories in the world where a complete multiphoton confocal microscope is available in a biosafety level (BSL)-3 environment.
Super Resolution Microscopy
Our facility has now entered the world of super resolution with a co-operative agreement giving access to a Leica STED 3D instrument. Until now the smallest objects one could resolve by conventional optical microscopy were 200nm across. Stimulated Emission Depletion (STED) microscopy allows the resolution limit to be lowered to approximately 65nm while still working with routine fluorescent probes.
Fluorescence Lifetime Imaging Microscopy (FLIM)
Protein-protein interactions have traditionally been shown by colocalization of probes, or by changes of intensity due to Förster resonance energy transfer (FRET). These measurements can be inaccurate due to changes in protein concentration or because of large pixel sizes. Fluorescence lifetime imaging is concentration independent, measuring the decay lifetimes of the fluorochromes rather than their intensities. A change in fluorescence lifetime as a result of molecular interaction can give direct measurements of binding affinities. The Biological Imaging Section (BIS) has five instruments equipped for FLIM measurements. FLIM data can be collected simultaneously with standard intensity images on the confocal systems.
Post-Collection Analysis
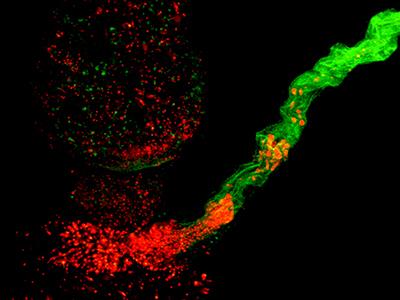
LysM-GFP mouse LN with B cells
Observation of the immune response to infectious disease is important to understanding of the underlying biological processes. The Biological Imaging Section has three state-of-the-art multiphoton imaging systems for intravital imaging. All three instruments are equipped with microscope enclosures for 37C incubation, as well as anesthesia for mice. Two of the instruments have dual IR lasers for excitation of probes across the IR spectrum. Additionally, the BIS is one of only a few laboratories in the world where a complete multiphoton confocal microscope is available in the biosafety level (BSL)-3 environment.
Additional Imaging Services
Poster Printing Service at Bethesda
The Biological Imaging facility will print posters for members of NIAID only.
Post-Collection Image Processing and Analysis
Facility staff can help with a wide range of image processing techniques such as deconvolution, image and movie adjustments and export for publication, protein interaction and colocalization, FLIM analysis, cell movement tracking and cell-cell interactions, tissue analysis, movie editing like joining, splitting and dimension adjustments, movie titling and compression, intensity and size/distance measurements, kymograph and cell time frames stabilization, gel blot and protein profile analysis etc.
- Imaris
- SVI Huygens Essentials
Computer Equipment Available for Use
There are 8 processing computers available for image processing needs. 8 computers are available in Building 4, one in building 33 (main campus) and one in Twinbrook 3. Computers are equipped with fast multiple core CPU and GPU with large amounts of RAM and storage capacity. Specific configurations for each computer and benchmark comparisons are listed here. Computer usage is free on first come first serve basis but if assistance is required, please schedule this in advance on this page.
Selected Publications
Rapid pathogen-specific recruitment of immune effector cells in the skin by secreted toxins.
Nguyen TH, Cheung GYC, Rigby KM, Kamenyeva O, Kabat J, Sturdevant DE, Villaruz AE, Liu R, Piewngam P, Porter AR, Firdous S, Chiou J, Park MD, Hunt RL, Almufarriji FMF, Tan VY, Asiamah TK, McCausland JW, Fisher EL, Yeh AJ, Bae JS, Kobayashi SD, Wang JM, Barber DL, DeLeo FR, Otto M. Nat Microbiol. 2022 Jan;7(1):62-72. doi: 10.1038/s41564-021-01012-9. Epub 2021 Dec 6. PMID: 34873293; PMCID: PMC8732318.
Metformin treatment rescues CD8+ T-cell response to immune checkpoint inhibitor therapy in mice with NAFLD.
Wabitsch S, McCallen JD, Kamenyeva O, Ruf B, McVey JC, Kabat J, Walz JS, Rotman Y, Bauer KC, Craig AJ, Pouzolles M, Phadke I, Catania V, Green BL, Fu C, Diggs LP, Heinrich B, Wang XW, Ma C, Greten TF. J Hepatol. 2022 Apr 1:S0168-8278(22)00178-7. doi: 10.1016/j.jhep.2022.03.010. Epub ahead of print. PMID: 35378172.
IBEX: an iterative immunolabeling and chemical bleaching method for high-content imaging of diverse tissues.
Radtke AJ, Chu CJ, Yaniv Z, Yao L, Marr J, Beuschel RT, Ichise H, Gola A, Kabat J, Lowekamp B, Speranza E, Croteau J, Thakur N, Jonigk D, Davis JL, Hernandez JM, Germain RN. Nat Protoc. 2022 Feb;17(2):378-401. doi: 10.1038/s41596-021-00644-9. Epub 2022 Jan 12. PMID: 35022622.
CD4 T cells are rapidly depleted from tuberculosis granulomas following acute SIV co-infection. Foreman TW, Nelson CE, Kauffman KD, Lora NE, Vinhaes CL, Dorosky DE, Sakai S, Gomez F, Fleegle JD, Parham M, Perera SR, Lindestam Arlehamn CS, Sette A; Tuberculosis Imaging Program, Brenchley JM, Queiroz ATL, Andrade BB, Kabat J, Via LE, Barber DL. Cell Rep. 2022 May 31;39(9):110896. doi: 10.1016/j.celrep.2022.110896. PMID: 35649361.
Mucus sialylation determines intestinal host-commensal homeostasis.
Yao Y, Kim G, Shafer S, Chen Z, Kubo S, Ji Y, Luo J, Yang W, Perner SP, Kanellopoulou C, Park AY, Jiang P, Li J, Baris S, Aydiner EK, Ertem D, Mulder DJ, Warner N, Griffiths AM, Topf-Olivestone C, Kori M, Werner L, Ouahed J, Field M, Liu C, Schwarz B, Bosio CM, Ganesan S, Song J, Urlaub H, Oellerich T, Malaker SA, Zheng L, Bertozzi CR, Zhang Y, Matthews H, Montgomery W, Shih HY, Jiang J, Jones M, Baras A, Shuldiner A, Gonzaga-Jauregui C, Snapper SB, Muise AM, Shouval DS, Ozen A, Pan KT, Wu C, Lenardo MJ. Cell. 2022 Mar 31;185(7):1172-1188.e28. doi: 10.1016/j.cell.2022.02.013. Epub 2022 Mar 17. PMID: 35303419; PMCID: PMC9088855.
Aedes aegypti Piwi4 Structural Features Are Necessary for RNA Binding and Nuclear Localization. Williams AE, Shrivastava G, Gittis AG, Ganesan S, Martin-Martin I, Valenzuela Leon PC, Olson KE, Calvo E. Int J Mol Sci. 2021 Nov 25;22(23):12733. doi: 10.3390/ijms222312733. PMID: 34884537; PMCID: PMC8657434.
Access
Currently, collaborating with RTB is only available to federally funded institutions. Researchers may access all RTB support services by visiting RTB on Inside NIAID (this link is only available to NIAID lab scientists).
Leadership
Owen M. Schwartz, Ph.D.
Chief, Biological Imaging Section

Owen M. Schwartz, Ph.D.