Mast Cell Biology Section
Established in 1995
Dean D. Metcalfe, M.D., M.S.
Special Volunteer
Scientist Emeritus
Specialty(s): Allergy and Immunology, Internal Medicine, Rheumatology Provides direct clinical care to patients at NIH Clinical Center

Major Areas of Research
- Identification of mutations and polymorphisms in human disease that affect the mast cell compartment
- Characterization of key signaling pathways in human mast cells that control mast cell responses
- Application of this information to the diagnosis and treatment of allergic and immunologic diseases and clonal mast cell disorders
Program Description
The mast cell is the focus of the Mast Cell Biology Section (MCBS) research effort. This multifunctional inflammatory cell is involved in both innate and acquired immunity and plays a central role in the induction of allergic inflammation. An integrated program investigating mast cell biology includes studies into the growth and differentiation of mast cells, mast-cell signal transduction, and the products generated by mast cells that lead to disease. The MCBS program emphasizes basic research that may be translated into the clinic and from the clinic to the bench, where protocols include studies on the pathogenesis of anaphylaxis, physical urticarias and clonal mast cell disorders. Research efforts have contributed to the identification of mutations in mast cell disease, understanding signaling through KIT and the high affinity IgE receptor, and how alterations in the control of mast cell mediator production affect human disease.
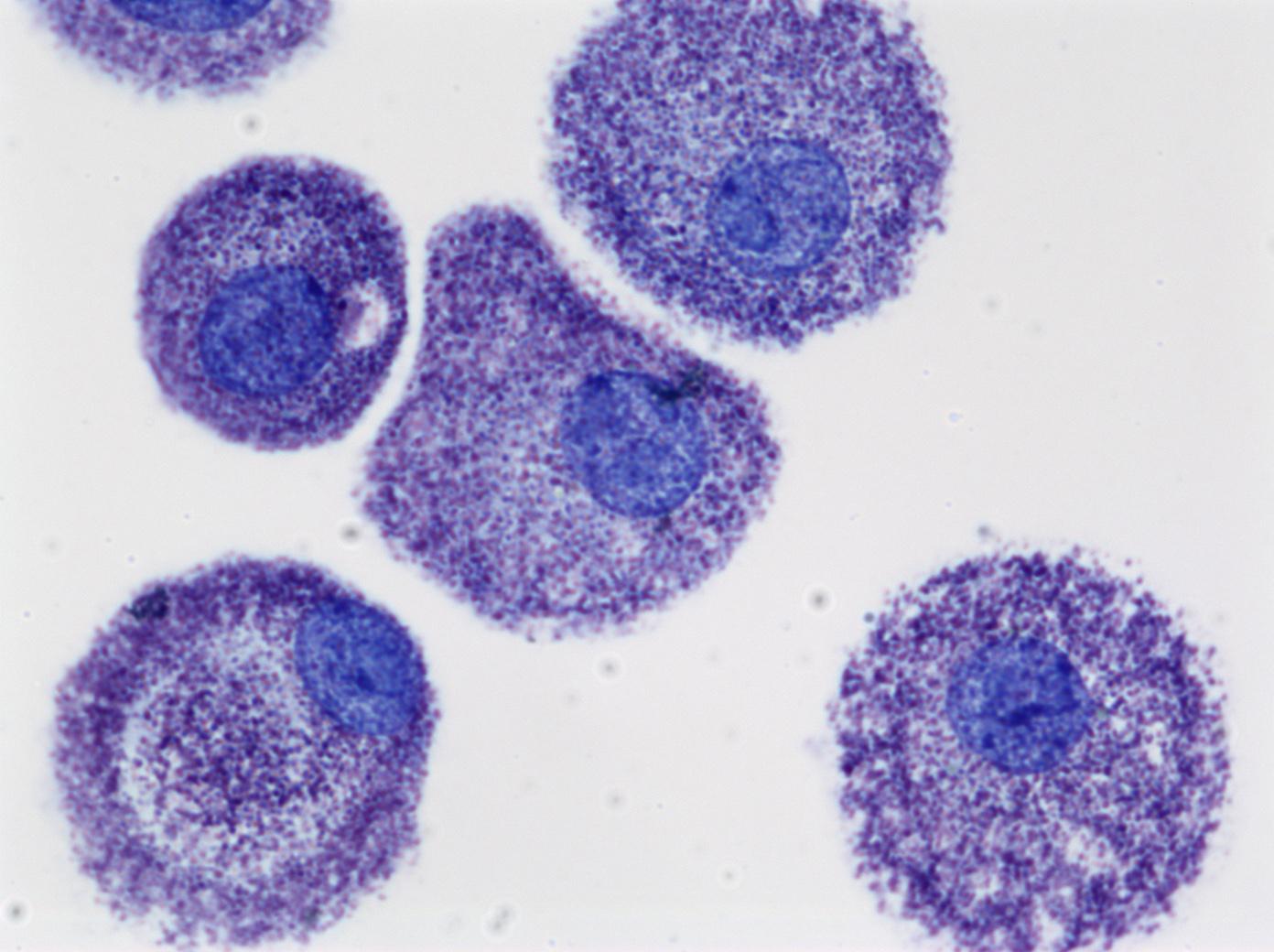
Cultured human mast cells. Credit: NIAID
Biography
Education
M.D., University of Tennessee
M.S., University of Michigan
Dr. Metcalfe received his M.D. at the University of Tennessee and an M.S. in microbiology at the University of Michigan, where he also did a residency in internal medicine. Dr. Metcalfe then trained in allergy and immunology during a fellowship at NIAID, followed by training in rheumatology while a Fellow in Immunology at the Robert Brigham Hospital in Boston. In 1995, he was appointed as the first Chief of the newly created Laboratory of Allergic Diseases at NIAID, a position he continued for 22 years until stepping down as Laboratory Chief in 2017. He is a past president of the American Academy of Allergy, Asthma, and Immunology, and a past chair of the American Board of Allergy and Immunology. Dr. Metcalfe is a Fellow of the American Academy of Allergy, Asthma, and Immunology and a member of the Association of American Physicians, Collegium Internationale Allergologicum, and American Clinical and Climatological Association. Dr. Metcalfe is a recipient of numerous awards including the Commendation, Outstanding Service and Meritorious Service Medals of the USPHS, an Outstanding Alumnus Award from University of Tennessee College of Medicine Alumni, the Distinguished Scientist Award from the Association the American Academy of Allergy and Asthma and Immunology, the Distinguished Scientist Award from the World Allergy Organization, the Lifetime Mentorship Award from the AAAAI and the ECNM Researcher of the Year.
Clinical Studies
Safety and Efficacy of Subcutaneous Sarilumab in Improving the Quality of Life in People With Indolent Systemic Mastocytosis - NCT03770273; Key words: Mastocytosis, sarilumab, IL-6
Study of Factors Regulating Mast Cell Proliferation - NCT00044122; Key words: Mastocytosis, clonal mast cell disorders, urticaria pigmentosa, cutaneous mastocytosis
Pathogenesis of Physical Induced Urticarial Syndromes - NCT00887939; Key words: Urticaria, hives, cholinergic, solar, vibratory
Cause of Unexplained Anaphylaxis - NCT00719719; Key words: anaphylaxis, shock, systemic allergic reaction
Selected Publications
Yin Y, Bai Y, Olivera A, Metcalfe DD. Demonstration and implications of IL-3 upregulation of CD25 expression on human mast cells. J Allergy Clin Immunol. 2022 Apr;149(4):1392-1401.e6.
Kim DK, Bandara G, Cho YE, Komarow HD, Donahue DR, Karim B, Baek MC, Kim HM, Metcalfe DD, Olivera A. Mastocytosis-derived extracellular vesicles deliver miR-23a and miR-30a into pre-osteoblasts and prevent osteoblastogenesis and bone formation. Nat Commun. 2021 May 5;12(1):2527.
Carter MC, Maric I, Brittain EH, Bai Y, Lumbard K, Bolan H, Cantave D, Scott LM, Metcalfe DD. A randomized double-blind, placebo-controlled study of omalizumab for idiopathic anaphylaxis. J Allergy Clin Immunol. 2021 Mar;147(3):1004-1010.e2.
Lyons JJ, Chovanec J, O'Connell MP, Liu Y, Šelb J, Zanotti R, Bai Y, Kim J, Le QT, DiMaggio T, Schwartz LB, Komarow HD, Rijavec M, Carter MC, Milner JD, Bonadonna P, Metcalfe DD, Korošec P. Heritable risk for severe anaphylaxis associated with increased α-tryptase-encoding germline copy number at TPSAB1. J Allergy Clin Immunol. 2021 Feb;147(2):622-632.
Carter MC, Bai Y, Ruiz-Esteves KN, Scott LM, Cantave D, Bolan H, Eisch R, Sun X, Hahn J, Maric I, Metcalfe DD. Detection of KIT D816V in peripheral blood of children with manifestations of cutaneous mastocytosis suggests systemic disease. Br J Haematol. 2018 Dec;183(5):775-782.
Kim DK, Cho YE, Komarow HD, Bandara G, Song BJ, Olivera A, Metcalfe DD. Mastocytosis-derived extracellular vesicles exhibit a mast cell signature, transfer KIT to stellate cells, and promote their activation. Proc Natl Acad Sci U S A. 2018 Nov 6;115(45):E10692-E10701.
Research Group
An integrated program investigating mast cell biology includes studies into growth & differentiation of mast cells, mast-cell signal transduction, & products generated by mast cells that lead to disease. MCBS emphasizes basic research that may be translated to the clinic & the bench. Protocols include studies on the pathogenesis of anaphylaxis, physical urticarias & clonal mast cell disorders.


