Microbial Pathogenesis Section
Mahtab Moayeri, Ph.D.
Associate Scientist, Microbial Pathogenesis Section
Major Areas of Research
- Inflammasome activation and the innate immune response
- Host factors controlling inflammasome-mediated events
- Role of the anthrax lethal and edema toxins in Bacillus anthracis pathogenesis
- Vaccines and therapeutics against Bacillus anthracis
Program Description
Bacillus anthracis produces two toxins with a common receptor binding component. Edema toxin (ET) is an adenylate cyclase. Lethal toxin (LT) is a protease that cleaves several mitogen-activated protein kinase kinases (MEKs/MKKs) and the regulatory subunits of phosphoinositide 3-kinase (p85-alpha, p85-beta), impacting signaling pathways which are central to cell survival, proliferation, stress response, metabolism and other essential functions. LT also cleaves and activates rodent inflammasome sensor NLRP1, resulting in downstream caspase-1 and gasdermin cleavage, followed by rapid cell death (pyroptosis).
Caspase-1 activation, which also occurs through activation of other inflammasome sensors, including the NLRP3, NAIP/NLRC4, and AIM2 sensors, initiates an innate immune response through maturation and release of the pro-inflammatory cytokines IL-1β and IL-18. Rat NLRP1 is also activated by Toxoplasma gondii, and controls disease sensitivity in this mode, but the mechanism for activation is currently unknown. My primary interests are understanding the signaling pathways targeted by anthrax toxins in cell and animal models. Our studies on anthrax toxin activation of NLRP1, shutdown of MEK signaling, modulation of PI3K/Akt signaling and dysregulation of cAMP signaling provide insight into disease pathogenesis, as well as aid in better understanding these important signaling pathways. Identification of novel toxin targets, new PAMPs and DAMPs which can activate the NLRP1 inflammasome, host genetic factors which control inflammasome-mediated responses, and anti-inflammatory or anti-anthrax therapeutics are areas of active study.
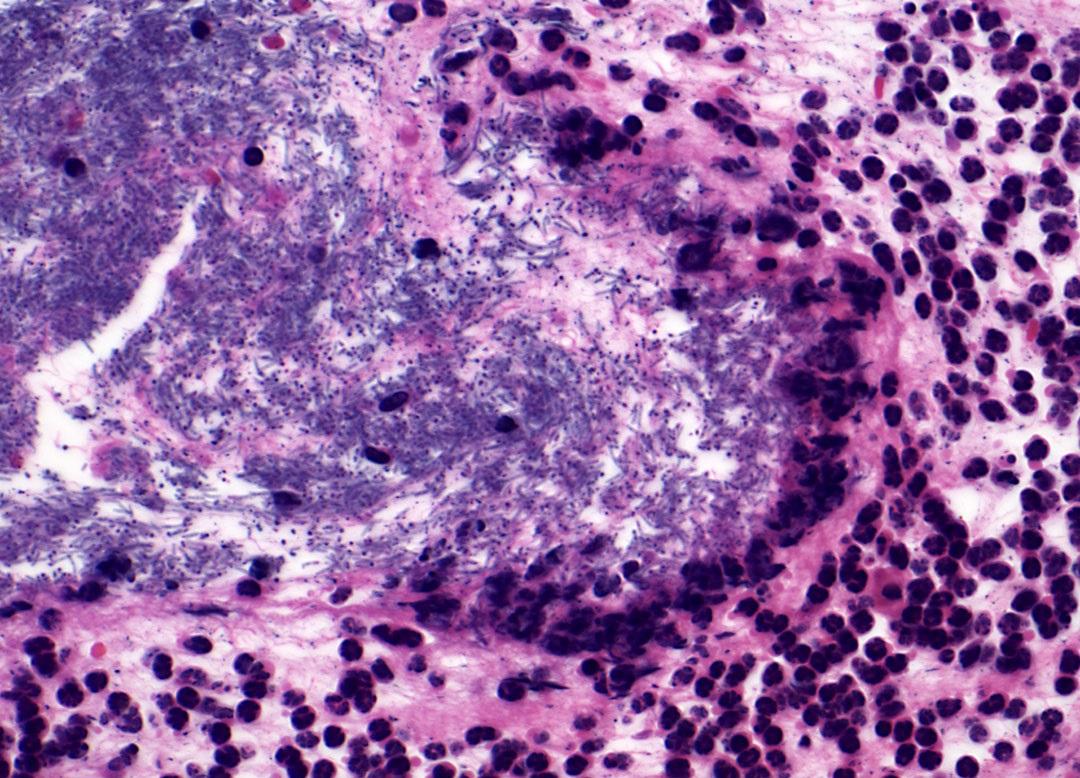
Bacillus anthracis spores and germinated vegetative bacterium vs. the innate immune response in mouse skin. Credit: NIAID
Biography
Education
Ph.D., University of Wisconsin-Madison
Dr. Moayeri completed her Ph.D. in Medical Microbiology and Immunology at the University of Wisconsin-Madison. She has focused on anthrax toxin pathogenesis research as a postdoctoral fellow, staff scientist and associate staff scientist at NIH since 1999.
Selected Publications
Merritt C, Chun EM, Fattah RJ, Silva LM, Ma QQ, Moayeri M, Paliga D, Neumann S, Heumann R, Leppla SH, Bugge TH. Imaging of anthrax intoxication in mice reveals shared and individual functions of surface receptors CMG-2 and TEM-8 in cellular toxin entry. J Biol Chem. 2022 Jan;298(1):101467.
Mendenhall MA, Liu S, Portley MK, O'Mard D, Fattah R, Szabo R, Bugge TH, Khillan JS, Leppla SH, Moayeri M. Anthrax lethal factor cleaves regulatory subunits of phosphoinositide-3 kinase to contribute to toxin lethality. Nat Microbiol. 2020 Dec;5(12):1464-1471.
Greaney AJ, Portley MK, O'Mard D, Crown D, Maier NK, Mendenhall MA, Mayer-Barber KD, Leppla SH, Moayeri M. Frontline Science: Anthrax lethal toxin-induced, NLRP1-mediated IL-1β release is a neutrophil and PAD4-dependent event. J Leukoc Biol. 2020 Sep;108(3):773-786.
Greaney AJ, Maier NK, Leppla SH, Moayeri M. Sulforaphane inhibits multiple inflammasomes through an Nrf2-independent mechanism. J Leukoc Biol. 2016 Jan;99(1):189-99.
Vrentas CE, Moayeri M, Keefer AB, Greaney AJ, Tremblay J, O'Mard D, Leppla SH, Shoemaker CB. A Diverse Set of Single-domain Antibodies (VHHs) against the Anthrax Toxin Lethal and Edema Factors Provides a Basis for Construction of a Bispecific Agent That Protects against Anthrax Infection. J Biol Chem. 2016 Oct 7;291(41):21596-21606.
Moayeri M, Leppla SH, Vrentas C, Pomerantsev AP, Liu S. Anthrax Pathogenesis. Annu Rev Microbiol. 2015;69:185-208.

