Helminth Immunology Section
Established in 1959
Thomas Nutman, M.D.
Lab Chief, Laboratory of Parasitic Diseases
Chief, Helminth Immunology Section
Provides direct clinical care to patients at NIH Clinical Center

Major Areas of Research
- Regulation of the host immune response to parasitic helminth infection (primarily filarial infections and those caused by soil-transmitted helminths)
- Influence of helminth infection on expression of non-parasitic infections, atopy, and asthma
- Molecular characterization of tissue-invasive helminth parasites
- Type 2-associated responses and their control in parasitic helminth infections and related disorders
- Clinical definition and pathogenesis underlying parasitic diseases
- New therapeutic interventions and methods of diagnosis in parasitic infections
Program Description
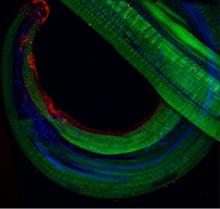
Surface expression (in red) of a filarial parasite-encoded molecule responsible for immune evasion in humans. Actin filaments are stained in green and nuclei in blue.
The focus of the Helminth Immunology Section is the study of host resistance and immune regulation in parasitic helminth infections of global importance. The ultimate goal of this work is prevention of infection and disease. Our work focuses on both the host response to helminth infection and the molecular basis for parasitism in helminths and the prototypical responses they induce.
Much of our work involves the analysis of host-parasite interaction using in vitro systems and studies of cells from infected patients ex vivo. Current activities include 1) functional mapping of the earliest host-parasite interaction that influences the polarized immune responses that are the hallmarks of these infections; 2) genomic and proteomic definition of each of the filarial parasites to identify parasite-encoded therapeutic, diagnostic, and vaccine-related targets; 3) studies of pathogenesis underlying disease manifestations (e.g., elephantiasis) in filarial infections; 4) studies of immunologic bystander effects of chronic helminth infection on non-parasitic infections (HIV, tuberculosis, malaria), atopy, and autoimmune diseases; 5) the regulation of igE and eosinophilia in the context of human helminth infections.
Biography
Education
A.B., Brown University, Providence, RI
M.D., University of Cincinnati College of Medicine, Cincinnati, OH
Dr. Nutman received his A.B. from Brown University and his M.D. from the University of Cincinnati College of Medicine. He did an internal medicine residency at New York University (Bellevue) and postdoctoral training in the Laboratory of Parasitic Diseases (LPD). He is board certified in internal medicine and allergy and immunology. He also holds a diploma/certificate in tropical medicine and travelers’ health. He has been at NIH in the Laboratory of Parasitic Diseases since 1982, where he is currently LPD's chief, as well as chief of the Helminth Immunology Section. In addition, he is the director of the NIAID International Center for Excellence in Research (ICER) located in Chennai, India, as well as director of the filariasis unit at the NIAID ICER in Mali. He is on numerous advisory committees and editorial boards and holds patents related to parasite diagnosis and vaccine development. He is the author or coauthor of over 600 papers and book chapters and has received multiple awards for his work in tropical medicine and immunology.
Selected Publications
Sears WJ, Qvarnstrom Y, Dahlstrom E, Snook K, Kaluna L, Baláž V, Feckova B, Šlapeta J, Modry D, Jarvi S, Nutman TB. AcanR3990 qPCR: A Novel, Highly Sensitive, Bioinformatically-Informed Assay to Detect Angiostrongylus cantonensis Infections. Clin Infect Dis. 2021 Oct 5;73(7):e1594-e1600.
Gazzinelli-Guimaraes PH, Bennuru S, de Queiroz Prado R, Ricciardi A, Sciurba J, Kupritz J, Moser M, Kamenyeva O, Nutman TB. House dust mite sensitization drives cross-reactive immune responses to homologous helminth proteins. PLoS Pathog. 2021 Mar 2;17(3):e1009337.
Ricciardi A, Nutman TB. IL-10 and Its Related Superfamily Members IL-19 and IL-24 Provide Parallel/Redundant Immune-Modulation in Loa loa Infection. J Infect Dis. 2021 Feb 3;223(2):297-305.
Easton A, Gao S, Lawton SP, Bennuru S, Khan A, Dahlstrom E, Oliveira RG, Kepha S, Porcella SF, Webster J, Anderson R, Grigg ME, Davis RE, Wang J, Nutman TB. Molecular evidence of hybridization between pig and human Ascaris indicates an interbred species complex infecting humans. Elife. 2020 Nov 6;9:e61562.
Ricciardi A, Bonne-Année S, Sciurba J, Karmele EP, Fujiwara RT, Nutman TB. Allergen presensitization drives an eosinophil-dependent arrest in lung-specific helminth development. J Clin Invest. 2019 Aug 5;129(9):3686-3701.
Kamgno J, Pion SD, Chesnais CB, Bakalar MH, D'Ambrosio MV, Mackenzie CD, Nana-Djeunga HC, Gounoue-Kamkumo R, Njitchouang GR, Nwane P, Tchatchueng-Mbouga JB, Wanji S, Stolk WA, Fletcher DA, Klion AD, Nutman TB, Boussinesq M. A Test-and-Not-Treat Strategy for Onchocerciasis in Loa loa-Endemic Areas. N Engl J Med. 2017 Nov 23;377(21):2044-2052.
Patents
Nutman TB, Bennuru S, Drame PM, inventors; The United States of America as represented by the Secretary, Department of Health and Human Services, assignee. Compositions and methods for detecting Loa loa. United States patent US 10,598,655. 24 Mar 2020.
Mitre EE, Morris C, Bennuru S, Nutman TB, inventors; The Henry M. Jackson Foundation for the Advancement of Military Medicine, Inc., The United States Government, as represented by the Secretary of the Department of Health and Human Services, assignees. Immunogenic compositions and vaccines for preventing or treating filarial disease. United states patent US 10,441,642. 15 Oct 2019.
Nutman TB, Fink DL, Burbelo PD, Kubofcik J, inventors; The United States of America as represented by the Secretary, Department of Health and Human Services, assignee. Diagnostic assays and methods of use for detection of filarial infection. U.S. application 61/410,239. 04 Nov 2010.
Nutman TB, Abraham D, inventors; The United States of America as represented by the Secretary, Department of Health and Human Services, assignee. Vaccine and methods of use against Strongyloides stercoralis infection. World patent WO/2011/097216. 11 Aug 2011.
Lazzerio MESL, Nutman TB, Weiss N, inventors; The United States of America, assignee. Nucleotide molecule encoding a specific Onchocerca volvulus antigen for the immunodiagnosis of onchocerciasis. United States patent US 5,416,009. 16 May 1995.
Visit the U.S. Patent and Trademark Office for a complete patent listing.
Research Group
The focus of the Helminth Immunology Section is the study of host resistance and immune regulation in parasitic helminth infections of global importance. The ultimate goal of this work is prevention of infection and disease. Our work focuses on both the host response to helminth infection and the molecular basis for parasitism in helminths and the prototypical responses they induce.


