Lymphocyte Activation Section
Susan Pierce, Ph.D.
Chief, Lymphocyte Activation Section

Program Description
The B-cell receptor (BCR) serves two essential functions in B-cell activation, namely the initiation of signaling cascades that lead to the transcription of a variety of genes associated with B-cell activation and the transport of antigen to intracellular compartments, where the antigen is processed and presented on MHC class II molecules for recognition by helper T cells. Provided with the appropriate T-cell help following antigen contact, B cells proliferate and differentiate into short-lived, antibody-secreting plasma cells as well as long-lived plasma cells and memory B cells that constitute immunological memory. The long-range goal of the Lymphocyte Activation Section (LAS) is to gain an understanding of the cellular and molecular mechanisms that underlie the initiation of BCR signaling, the intracellular trafficking of the BCR, and the generation, maintenance, and activation of B-cell memory. Of particular interest is the regulation of these processes by B cell-activating and inhibitory co-receptors and during immune responses to infection. The long range goals of LAS are as follows:
- Gain an understanding of the molecular mechanisms by which antigen engagement by BCR triggers a signaling cascade. A key element of this aim is the use of live-cell imaging techniques that allows analyses of the earliest events in the initiation of signaling that follow within seconds of the binding of the antigen to BCR at the single molecule level.
- Determine the mechanisms that underlie the internalization and trafficking of BCR to MHC class II-containing intracellular compartments. These studies are defining the spatial relationship between the location of the BCR and the activation of various components of the signaling cascade, including the location of the interactions of the BCR with the intracellular innate immune system Toll-like receptors expressed by B cells that modulate BCR signaling.
- Define the mechanisms that underlie the regulation of B-cell responses by the innate immune system receptors, including the Toll-like receptors, the activating receptor complex CD19/CD21, and the inhibitory receptor FcγRIIB.
- Gain an understanding of the cellular mechanisms underlying the generation, maintenance, and activation of B cell immunological memory. These studies are focused on the acquisition and maintenance of memory in response to antigens of the malaria parasite Plasmodium falciparum, in response to vaccination in the United States and to natural infection in Africa.
It is hoped that knowledge gained through these studies will add fundamentally to our understanding of B-cell activation and its regulation. Such knowledge is likely to aid research efforts in two areas of high public health priority: namely, the development of new therapeutics to control B-cell responses in autoimmune disease and the design of effective vaccines to control infectious diseases.
Imaging the earliest events in BCR signaling and trafficking in living B cells in real time

Figure 1: The total internal reflection (TIRF) image of a B cell expressing fluorescent BCR as it engages a fluid lipid bilayer that contains the B cell antigen. The view is looking up at the B cell showing BCRs as the B cell contacts the bilayer, spreads and then actively moves BCRs to form a synapse.
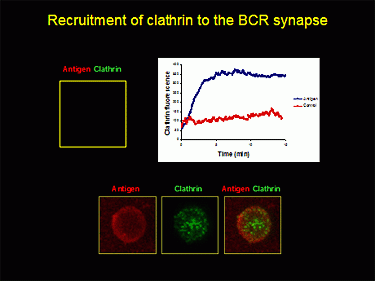
Figure 2: A TIRF image of the recruitment of clatherin (in green) from the cytosol to the synapse as the BCR internalizes antigen (in red) into the cell.
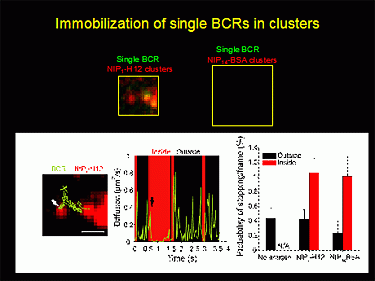
Figure 3: Single molecule images of individual BCR (green) as they stop when encounter BCR-antigen clusters (red).
Understanding the mechanisms underlying synergy in signaling between adaptive and innate immune receptors
Figure 4: A confocal image demonstrating the recruitment of TLR9 to the intracellular compartment where the BCR is internalized from the surface. Synergistic signaling to the MAP kinase occurs in this compartment.

Figure 5: Two volunteers in our study.
Figure 6: A proteomic microarray containing proteins from approximately 25 percent of thePlasmodium falciparum genome probed with sera from volunteers in our study sorted by those who had malaria and those that did not. The protein microarray is one of several immune parameters we are assessing.
Figures 5 and 6: We are carrying out a longitudinal study in Mali, Africa, to determine the effect of malaria infections on the acquisition of immunity to malaria.
Biography
Education
Ph.D., 1976, University of Pennsylvania, Philadelphia, PA
Dr. Pierce became chief of the NIAID Laboratory of Immunogenetics in 1999. Prior to joining NIAID, she was a member of the faculty at Northwestern University, where she held the Cook Chair in the Biological Sciences. She earned her Ph.D. in immunology from the University of Pennsylvania in 1976.
Selected Publications
Waisberg M, Lin CK, Huang CY, Pena M, Orandle M, Bolland S, Pierce SK. The impact of genetic susceptibility to systemic lupus erythematosus on placental malaria in mice. PLoS One. 2013 May 10;8(5):e62820.
Liu W, Chen E, Zhao XW, Wan ZP, Gao YR, Davey A, Huang E, Zhang L, Crocetti J, Sandoval G, Joyce MG, Miceli C, Lukszo J, Aravind L, Swat W, Brzostowski J, Pierce SK. The scaffolding protein synapse-associated protein 97 is required for enhanced signaling through isotype-switched IgG memory B cell receptors. Sci Signal. 2012 Jul 31;5(235):ra54.
Waisberg M, Cerqueira GC, Yager SB, Francischetti IM, Lu J, Gera N, Srinivasan P, Miura K, Rada B, Lukszo J, Barbian KD, Leto TL, Porcella SF, Narum DL, El-Sayed N, Miller LH, Pierce SK. Plasmodium falciparum merozoite surface protein 1 blocks the proinflammatory protein S100P. Proc Natl Acad Sci U S A. 2012 Apr 3;109(14):5429-34.
Davey AM, Pierce SK. Intrinsic differences in the initiation of B-cell receptor signaling favor responses of human IgG+ memory B cells over IgM+ naive B cells. J Immunol. 2012 Apr 1;188(7):3332-41.
Davey AM, Liu W, Sohn HW, Brzostowski J, Pierce SK. Understanding the initiation of B-cell signaling through live cell imaging.Methods Enzymol. 2012;506:265-90.
Waisberg M, Vickers BK, Yager SB, Lin CK, Pierce SK. Testing in mice the hypothesis that melanin is protective in malaria infections. PLoS One. 2012;7(1):e29493.

