Molecular Microbiology Section
Established in 1995
K.J. Kwon-Chung, Ph.D.
Chief, Molecular Microbiology Section, LCIM

Major Areas of Research
- Virulence determinants of Cryptococcus neoformans and Aspergillus fumigatus
- Mechanism by which Cryptococcus neoformans invades the brain
- Mechanism by which cryptococci adapt to the brain environment and produce fulminating disease
- The signaling mechanism involved in the cryptococcal adaptation to human brain environment
- The mechanism of cryptococcal adaptive resistance to azole drugs and flucytosine
- Identification of pathobiological differences between Cryptococcus neoformans and its sibling species, Cryptococcus gattii
- Identification of host factors that predispose patients to invasive aspergillosis
Program Description
The Molecular Microbiology Section is focused on the pathobiology of Cryptococcus neoformans and Aspergillus fumigatus, two of the most common and serious fungal pathogens that primarily affect immunocompromised patients. Both C. neoformans and A. fumigatus are environmental pathogens that cause life-threatening systemic disease upon inhalation. The fatality rate of these diseases is high even if treated with the most effective antimycotic agents currently available.
Cryptococcus neoformans
Cryptococcus neoformans, an environmental yeast species most commonly found in pigeon droppings, tree barks, and soil contaminated with avian guano, is the etiologic agent of cryptococcosis. Cryptococcosis has been recognized as an AIDS-defining disease because the major risk group of the disease is people infected with HIV. Although C. neoformans can cause lesions in almost every organ after being inhaled by the host, the most common clinical manifestation and most common cause of death is meningoencephalitis, an infection of the brain. Cryptococcus gattii, a sibling species of C. neoformans, also causes cryptococcosis but more frequently in otherwise healthy patients without well defined underlying risks. The two species share 85 to 90 percent genomic sequence homology but differ significantly in their ecology, epidemiology, and biochemistry. Our section is currently focusing on their differences in pathobiology as well as the way the host responds to the two pathogens.
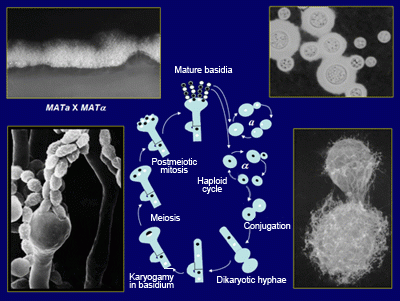
Cryptococcus neoformans Lifecycle
Pathogenic Aspergillus species are ubiquitous in our environment, and humans regularly inhale numerous Aspergillus spores (conidia) during daily life. Of those species, Aspergillus fumigatus is known to be the major cause of life-threatening invasive pulmonary aspergillosis (IPA) in immunocompromised patients throughout the world. The major risk for IPA is profound neutropenia and chronic granulomatous disease (CGD). However, more than 50 percent of CGD patients, as well as neutropenic patients, do not experience IPA in their lifetime. The Molecular Microbiology Section is currently focused on the pathobiological differences between multiple sibling species that belong to Aspergillus Section Fumigati and host genetic factors that predispose patients to IPA.
Biography
Education
Ph.D., University of Wisconsin, Madison
B.S., M.S., Ewha Womans University, Seoul, South Korea
Dr. Kwon-Chung received her B.S. and M.S. in biology from Ewha Womans University in Seoul, South Korea, prior to receiving a Fulbright Scholarship to pursue her doctoral work in the Department of Bacteriology at the University of Wisconsin, Madison. After receiving her Ph.D. in 1965, she joined the Medical Mycology Section of the NIAID Laboratory of Microbiology in 1966 as a visiting fellow. She became a senior investigator in the NIAID Laboratory of Clinical Investigation in 1973 and has been the chief of the Molecular Microbiology Section, Laboratory of Clinical Immunology and Microbiology, since 1995. She received Honorary Doctoral Degree in Science from University of Wisconsin in 2009 and the Lifetime Achievement Award from the American Society for Microbiology in 2017.
Selected Publications
Yang DH, England MR, Salvator H, Anjum S, Park YD, Marr KA, Chu LA, Govender NP, Lockhart SR, Desnos-Ollivier M, Chen S, Halliday C, Kan A, Chen J, Wollenberg KR, Zelazny A, Perfect JR, Chang YC, Bennett JE, Holland SM, Meyer W, Williamson PR, Kwon-Chung KJ. Cryptococcus gattii Species Complex as an Opportunistic Pathogen: Underlying Medical Conditions Associated with the Infection. mBio. 2021 Oct 26;12(5):e0270821.
Davis MJ, Moyer S, Hoke ES, Sionov E, Mayer-Barber KD, Barber DL, Cai H, Jenkins L, Walter PJ, Chang YC, Kwon-Chung KJ. Pulmonary Iron Limitation Induced by Exogenous Type I IFN Protects Mice from Cryptococcus gattii Independently of T Cells. mBio. 2019 Jun 18;10(3):e00799-19.
Seyedmousavi S, Davis MJ, Sugui JA, Pinkhasov T, Moyer S, Salazar AM, Chang YC, Kwon-Chung KJ. Exogenous Stimulation of Type I Interferon Protects Mice with Chronic Granulomatous Disease from Aspergillosis through Early Recruitment of Host-Protective Neutrophils into the Lung. mBio. 2018 Mar 27;9(2):e00422-18.
Chang YC, Khanal Lamichhane A, Kwon-Chung KJ. Cryptococcus neoformans, Unlike Candida albicans, Forms Aneuploid Clones Directly from Uninucleated Cells under Fluconazole Stress. mBio. 2018 Dec 4;9(6):e01290-18.
Kwon-Chung KJ, Bennett JE, Wickes BL, Meyer W, Cuomo CA, Wollenburg KR, Bicanic TA, Castañeda E, Chang YC, Chen J, Cogliati M, Dromer F, Ellis D, Filler SG, Fisher MC, Harrison TS, Holland SM, Kohno S, Kronstad JW, Lazera M, Levitz SM, Lionakis MS, May RC, Ngamskulrongroj P, Pappas PG, Perfect JR, Rickerts V, Sorrell TC, Walsh TJ, Williamson PR, Xu J, Zelazny AM, Casadevall A. The Case for Adopting the "Species Complex" Nomenclature for the Etiologic Agents of Cryptococcosis. mSphere. 2017 Jan 11;2(1):e00357-16.
Losada L, Sugui JA, Eckhaus MA, Chang YC, Mounaud S, Figat A, Joardar V, Pakala SB, Pakala S, Venepally P, Fedorova N, Nierman WC, Kwon-Chung KJ. Genetic Analysis Using an Isogenic Mating Pair of Aspergillus fumigatus Identifies Azole Resistance Genes and Lack of MAT Locus's Role in Virulence. PLoS Pathog. 2015 Apr 24;11(4):e1004834.
Research Group
Studies on pathobiology of medically important fungi especially of those causing cryptococcosis and aspergillosis.


